Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (33): 5396-5403.doi: 10.3969/j.issn.2095-4344.1840
Previous Articles Next Articles
Satellite glial cells in dorsal root ganglia: implication for pain regulation
Zhang Xiaozhuo1, Lü Tingting1, Wang Zhaoqin1, Weng Zhijun2, Cui Yunhua2, Zhang Fang2, Zhao Min2, Liu Huirong2, Wu Huangan2
- 1Yueyang Clinical College, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China; 2Shanghai Research Institute of Acupuncture and Meridian, Shanghai 200030, China
-
Revised:2019-06-18Online:2019-11-28Published:2019-11-28 -
Contact:Cui Yunhua, MD, Associate researcher, Master’s supervisor, Shanghai Research Institute of Acupuncture and Meridian, Shanghai 200030, China -
About author:Zhang Xiaozhuo, Master candidate, Yueyang Clinical College, Shanghai University of Traditional Chinese Medicine, Shanghai 201203, China -
Supported by:the National Program on Key Basic Research Project of China, No. 2015CB554501 (to WHG); the National Natural Science Foundation of China (General Program), No. 81873367 (to WZJ); Shanghai Young Talents Sailing Plan, No. 18YF1421600 (to ZF); Scientific Research Topics of Shanghai Health and Planning Commission, No. 20174Y0015 (to ZF); Shanghai Leading Medical Talents Project (2015), No. 80 (to LHR); Shanghai Three-Year Plan for the Development of Traditional Chinese Medicine, No. ZY(2018-2020)-CCCX-2004-01 (to WHG)
CLC Number:
Cite this article
Zhang Xiaozhuo, Lü Tingting, Wang Zhaoqin, Weng Zhijun, Cui Yunhua, Zhang Fang, Zhao Min, Liu Huirong, Wu Huangan. Satellite glial cells in dorsal root ganglia: implication for pain regulation[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(33): 5396-5403.
share this article
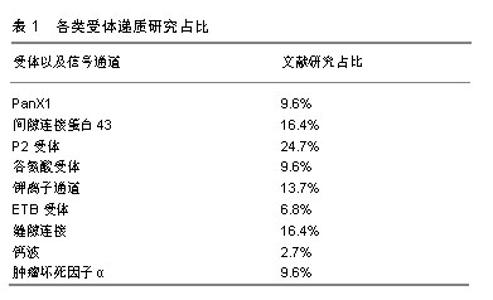
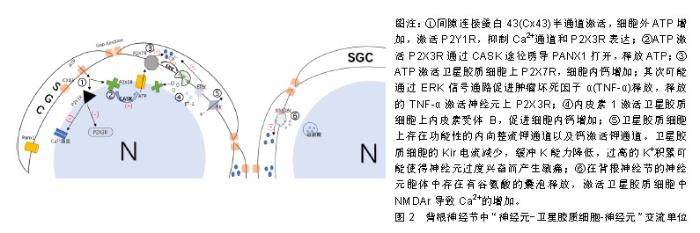
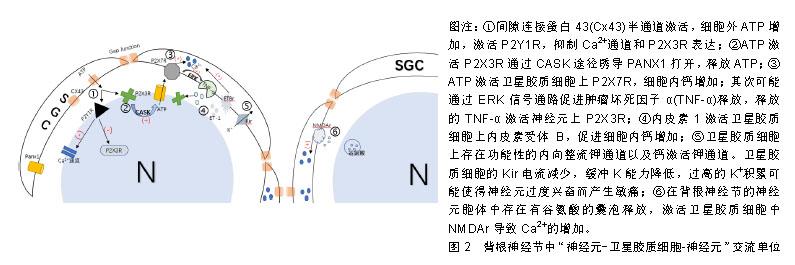
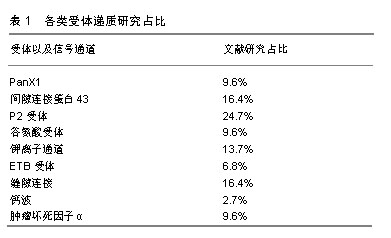
2.1 神经元与卫星胶质细胞的结构形态 背根神经节是感觉神经节的主要类型,是痛觉传入的第一站,支配着包括内脏在内的身体大部分部位的感觉传入。背根神经节中神经元外紧密的包绕着卫星胶质细胞,并与之形成了独特的功能单位[6-7],在卫星胶质细胞鞘膜外又有结缔组织环绕,把它与相邻的神经元鞘区分开[8],由此形成了由单个神经元与其鞘组成的独立单元,也有2个或2个以上神经元由共同胶质细胞覆盖,形成的神经细胞簇。卫星胶质细胞包绕的内部,神经元与卫星胶质细胞鞘膜之间间隔仅约20 nm,神经元外细胞空间非常小,而正是这种紧密的位置关系,有助于卫星胶质细胞控制神经元周围环境(亦即鞘内环境),也有助于神经元细胞与胶质细胞之间的信号传递[7],为了进一步增加这种信号传递,神经元可以发出一些细微凸起(微绒毛),与胶质细胞的内陷相吻合[9],进一步增加了神经元与卫星胶质细胞之间接触面积。 在卫星胶质细胞紧密包绕下,神经元与外界相隔离,背根神经节有机汞中毒的试验中也证明了此点[10],无论剂量高低,大多数汞都出现在神经元周围的胶质细胞中,神经元细胞标记不太明显。而卫星胶质细胞被有机汞标记后开始出现细胞增殖和肥大,胶质细胞的变化为神经元提供了很好的的保护,一定程度减缓了这些分子的扩散。但是在许多研究中也发现,不仅仅是离子和小型神经递质,甚至大分子如蛋白质等都可渗透卫星胶质细胞鞘膜,进入神经细胞外间隙[8]。因此卫星胶质细胞不是神经元的绝对屏障,卫星胶质细胞对神经元有一定的保护作用,同时也允许部分小分子进入鞘膜实现对神经元的调节。 2.2 卫星胶质细胞-背根神经节之间的信号传递 背根神经节内神经元胞体和卫星胶质细胞都对ATP、细胞因子等递质敏感,且神经元与卫星胶质细胞表面分别主要表达不同的受体,如神经元表达P2X3受体,而卫星胶质细胞主要表达P2X7受体,各类受体递质研究占比见表1。而这些递质、受体也是卫星胶质细胞与神经元以及与邻近胶质细胞信号传递的主要方式。通过神经元将信息传递给卫星胶质细胞,卫星胶质细胞传递到邻近卫星胶质细胞,邻近胶质细胞再传递给鞘内神经元的方式实现神经元与神经元之间的交流,形成类似于“神经元-卫星胶质细胞-神经元”的交流单位(见图2)。"
| [1]Shim B , Kim DW , Kim BH , et al. Mechanical and heat sensitization of cutaneous nociceptors in rats with experimental peripheral neuropathy. Neuroscience. 2005; 132(1):193-201. [2]Devor M. Ectopic discharge in Abeta afferents as a source of neuropathic pain.Exp Brain Res. 2009; 196(1):115-128.[3]Ying M , Liu H , Zhang T , et al. Effect of artemisinin on neuropathic pain mediated by P2X4 receptor in dorsal root ganglia. Neurochem Int. 2017;108:27-33.[4]Hanani M. Satellite glial cells in sensory ganglia: from form to function. Brain Res Brain Res Rev.2005;48(3):457-476.[5]Souza GR, Talbot J, Lotufo CM, et al. Fractalkine mediates inflammatory pain through activation of satellite glial cells. Proc Natl Acad Sci U S A. 2013;110(27):11193-11198.[6]Hanani M. Role of satellite glial cells in gastrointestinal pain. Frontiers in Cellular.Neuroscience. 2015;9(7):412.[7]Hanani M. Intercellular communication in sensory ganglia by purinergic receptors and gap junctions: Implications for chronic pain. Brain Res.2012;1487(17):183-191.[8]Pannese E. The structure of the perineuronal sheath of satellite glial cells (SGCs) in sensory ganglia. Neuron Glia Biol.2010;6(1):3-10.[9]Pannese E. Perikaryal surface specializations of neurons in sensory ganglia. Int Rev Cytol. 2002;220:1-34.[10]Schiønning JD, Danscher G.Autometallographic mercury correlates with degenerative changes in dorsal root ganglia of rats intoxicated with organic mercury. APMIS. 1999;107(3): 303-310.[11]Procacci P , Magnaghi V , Pannese E. Perineuronal satellite cells in mouse spinal ganglia express the gap junction protein connexin43 throughout life with decline in old age. Brain Res Bull.2008;75(5):0-569.[12]Ohara PT, Vit JP, Bhargava A, et al. Evidence for a Role of Connexin 43 in Trigeminal Pain Using RNA Interference In Vivo. J Neurophysiol.2008;100(6):3064-3073.[13]Chen J, He L, Dinger B , et al. Chronic hypoxia upregulates connexin43 expression, in rat carotid body and petrosal ganglion. J Appl Physiol. 2002;92(4):1480-1486.[14]Penuela S, Gehi R, Laird DW.The biochemistry and function of pannexin channels. Biochim Biophys Acta. 2013;1828(1): 15-22.[15]Zhang Y, Laumet G , Chen S R , et al. Pannexin-1 Upregulation in the Dorsal Root Ganglion Contributes to Neuropathic Pain Development.J Biol Chem.2015;290(23):14647.[16]Montero TD, Orellana JA . Hemichannels: new pathways for gliotransmitter release.Neuroscience.2015; 286:45-59.[17]Retamal MA , Alcayaga J , Verdugo CA , et al. Opening of pannexin- and connexin-based channels increases the excitability of nodose ganglion sensory neurons.Front Cell Neurosci. 2014;8:158.[18]Kaji K, Shinoda M , Honda K , et al. Connexin 43 contributes to ectopic orofacial pain following inferior alveolar nerve injury. Mol Pain.2016;12:12/0/1744806916633704.[19]Retamal MA, Riquelme MA, Jimmy S, et al. Connexin43 Hemichannels in Satellite Glial Cells, Can They Influence Sensory Neuron Activity?. Front Mol Neurosci. 2017;10:374.[20]Chen G, Park CK , Xie RG , et al. Connexin-43 induces chemokine release from spinal cord astrocytes to maintain late-phase neuropathic pain in mice.Brain. 2014;137(Pt 8): 2193-2209.[21]Retamal MA, Reyes EP, Garc?-A IE,et al.Diseases associated with leaky hemichannels.Front Cell Neurosci. 2015;9:267.[22]Ohara PT, Vit JP , Bhargava A, et al. Evidence for a Role of Connexin 43 in Trigeminal Pain Using RNA Interference In Vivo. J Neurophysiol. 2008;100(6):3064-3073.[23]Jasmin L, Vit J P, Bhargava A , et al. Can satellite glial cells be therapeutic targets for pain control?. Neuron Glia Biol.2010; 6(1):63-71.[24]Xu Q , Cheong YK , Yang F , et al. Intrathecal carbenoxolone inhibits neuropathic pain and spinal wide-dynamic range neuronal activity in rats after an L5 spinal nerve injury. Neurosci Lett. 2014;563:45-50.[25]Bele T , Fabbretti E. The scaffold protein calcium/calmodulin‐dependent serine protein kinase controls ATP release in sensory ganglia upon P2X3 receptor activation and is part of an ATP keeper complex. J Neurochem. 2016;138(4):11.[26]Huang LYM, Gu Y, Chen Y. Communication between neuronal somata and satellite glial cells in sensory ganglia. Glia. 2013; 61(10):1571-1581.[27]Nakatsuka T, Gu JG. P2X purinoceptors and sensory transmission. Pslug Arch Eur J Ph. 2006;452(5):598-607.[28]Chen Y, Li G, Huang LY.P2X7 receptors in satellite glial cells mediate high functional expression of P2X3 receptors in immature dorsal root ganglion neurons. Mol Pain. 2012;8:9.[29]Franke H, Verkhratsky A, Burnstock G, et al. Pathophysiology of astroglial purinergic signalling. Purinerg Signal. 2012;8(3): 629-657.[30]Wu B, Ma Y, Yi Z . Resveratrol-decreased hyperalgesia mediated by the P2X7 receptor in gp120-treated rats.Mol Pain. 2017;13:1744806917707667.[31]Liu J, Li G, Peng H , et al. Sensory–sympathetic coupling in superior cervical ganglia after myocardial ischemic injury facilitates sympathoexcitatory action via P2X7receptor. Purinerg Signal.2013;9(3):463-479.[32]Chessell IP, Hatcher JP, Bountra C , et al. Disruption of the P2X7 purinoceptor gene abolishes chronic inflammatory and neuropathic pain. Pain.2005;114(3):386-396.[33]Song J, Ying Y, Wang W, et al. The role of P2X7R/ERK signaling in dorsal root ganglia satellite glial cells in the development of chronic postsurgical pain induced by skin/muscle incision and retraction (SMIR). Brain Behav Immun. 2017.[34]Xie J , Liu S , Wu B , et al. The protective effect of resveratrol in the transmission of neuropathic pain mediated by the P2X7 receptor in the dorsal root ganglia. Neurochem Int. 2017;103: 24-35.[35]Molliver DC, Malin SA. Gi- and Gq-coupled ADP (P2Y) receptors act in opposition to modulate nociceptive signaling and inflammatory pain behavior. Mol Pain.2010;6(1):1-12.[36]Magni G, Ceruti S. P2Y purinergic receptors: New targets for analgesic and antimigraine drugs. Biochem Pharmacol. 2013;85(4):466-477.[37]Yousuf A, Klinger F, Schicker K, et al. Nucleotides control the excitability of sensory neurons via two P2Y receptors and a bifurcated signaling cascade.Pain.2011;152(8): 1899-1908.[38]Chen Y, Zhang X, Wang C, et al. Activation of P2X7 receptors in glial satellite cells reduces pain through downregulation of P2X3 receptors in nociceptive neurons. Proc Natl Acad Sci U S A. 2008;105(43):16773-16778. [39]Gerevich Z, Christoph Müller, Illes P . Metabotropic P2Y1 receptors inhibit P2X3 receptor-channels in rat dorsal root ganglion neurons.Eur J Pharmacol.2005;521(1-3):34-38.[40]Gerevich Z, Borvendeg SJ, Schröder, Wolfgang, et al. Inhibition of N-type voltage-activated calcium channels in rat dorsal root ganglion neurons by P2Y receptors is a possible mechanism of ADP-induced analgesia.J Neurosci. 2004;24(4): 797-807.[41]Mo G , Peleshok J C , Cao C Q , et al. Control of P2X3 channel function by metabotropic P2Y2 utp receptors in primary sensory neurons. Mol Pharmacol. 2013;83(3): 640-647.[42]Castillo C , Norcini M , Hernandez L A M , et al. Satellite glia cells in dorsal root ganglia express functional NMDA receptors. Neuroscience.2013;240(9):135-146.[43]Gu Y, Chen Y, Zhang X, et al. Neuronal soma–satellite glial cell interactions in sensory ganglia and the participation of purinergic receptors. Neuron Glia Biol. 2010;6(01):10.[44]Ferrari LF , Lotufo CM , Dionéia Araldi, et al. Inflammatory sensitization of nociceptors depends on activation of NMDA receptors in DRG satellite cells. Proc Natl Acad Sci U S A. 2014;111(51):18363-18368.[45]Carozzi V A , Canta A , Oggioni N, et al. Expression and distribution of ‘high affinity’ glutamate transporters GLT1, GLAST, EAAC1 and of GCPII in the rat peripheral nervous system. J Anat.2008;213(5):8.[46]Ohara P, Vit J P , Bhargava A , et al. Gliopathic Pain: When Satellite Glial Cells Go Bad. Neuroscientist. 2009;15(5): 450-63.[47]Sung B , Lim G , Mao J . Altered expression and uptake activity of spinal glutamate transporters after nerve injury contribute to the pathogenesis of neuropathic pain in rats. J Neurosci.2003;23(7):2899.[48]Tang X , Schmidt TM , Perezleighton CE , et al. Inwardly rectifying potassium channel Kir4.1 is responsible for the native inward potassium conductance of satellite glial cells in sensory ganglia. Neuroscience. 2010;166(2):397-407.[49]Zhang H , Mei X , Zhang P , et al. Altered Functional Properties of Satellite Glial Cells in Compressed Spinal Ganglia. Glia.2009;57(15):1588-1599.[50]Silva JR, Lopes AH, Talbot J, et al. Neuro-immune-glia interactions in the sensory ganglia account for the development of acute herpetic neuralgia. J Neurosci. 2017; 37(27):6408-6422.[51]Takeda M , Takahashi M , Nasu M , et al. Peripheral inflammation suppresses inward rectifying potassium currents of satellite glial cells in the trigeminal ganglia.Pain.2011;152(9): 2147-2156.[52]Vit J P , Ohara PT , Bhargava A, et al. Silencing the Kir4.1 Potassium Channel Subunit in Satellite Glial Cells of the Rat Trigeminal Ganglion Results in Pain-Like Behavior in the Absence of Nerve Injury.J Neurosci.2008;28(16):4161-4171.[53]Murata Y, Yasaka T, Takano M , et al. Neuronal and glial expression of inward rectifier potassium channel subunits Kir2.x in rat dorsal root ganglion and spinal cord. Neurosci Lett. 2016;617:59-65.[54]Pagadala P, Park CK , Bang S, et al. Loss of NR1 Subunit of NMDARs in Primary Sensory Neurons Leads to Hyperexcitability and Pain Hypersensitivity: Involvement of Ca2+-Activated Small Conductance Potassium Channels. J Neurosci.2013;33(33):13425-13430.[55]Thompson JM, Ji G, Neugebauer V. Small-Conductance Calcium-Activated Potassium (SK) Channels in the Amygdala Mediate Pain-Inhibiting Effects of Clinically Available Riluzole in a Rat Model of Arthritis Pain. Mol Pain. 2015;11(1): s12990-015-0055.[56]Tsantoulas C , Mcmahon SB . Opening paths to novel analgesics: the role of potassium channels in chronic pain. Trends Neurosci 2014;37(3):146-158.[57]Lu R, Flauaus C, Kennel L, et al. K Ca 3.1 channels modulate the processing of noxious chemical stimuli in mice. Neuropharmacology.2017;125:386-395.[58]Pomonis JD, Rogers SD, Peters CM , et al. Expression and Localization of Endothelin Receptors: Implications for the Involvement of Peripheral Glia in Nociception. J Neurosci. 2001;21(3):999-1006.[59]Piovezan AP, D'Orléans-Juste P, Souza GE, et al. Endothelin-1-induced ET A, receptor-mediated nociception, hyperalgesia and oedema in the mouse hind-paw: modulation by simultaneous ET B, receptor activation.Br J Pharmacol. 2010;129(5):961-968.[60]Vellani V, Prandini M , Giacomoni C , et al. Functional endothelin receptors are selectively expressed in isolectin B4-negative sensory neurons and are upregulated in isolectin B4-positive neurons by neurturin and glia-derived neurotropic factor. Brain Res.2011;1381(none):31-37.[61]Feldman-Goriachnik R, Hanani M. Functional study of endothelin B receptors in satellite glial cells in trigeminal ganglia. NeuroReport.2011;22(10):465-469.[62]Feldman-Goriachnik R, Hanani M. The effects of endothelin-1 on satellite glial cells in peripheral ganglia. Neuropeptides. 2017;63:37-42.[63]Huang TY , Belzer V , Hanani M . Gap junctions in dorsal root ganglia: Possible contribution to visceral pain. Eur J Pain. 2010;4(1):490-2147483647.[64]Hanani M , Huang TY , Cherkas PS , et al. Glial cell plasticity in sensory ganglia induced by nerve damage. Neuroscience. 2002;114(2):279-283.[65]Dublin P, Hanani M . Satellite glial cells in sensory ganglia: Their possible contribution to inflammatory pain. Brain Behav Immun. 2007;21(5):592-598.[66]Liu W , Glueckert R , Linthicum F H , et al. Possible role of gap junction intercellular channels and connexin 43 in satellite glial cells (SGCs) for preservation of human spiral ganglion neurons. Cell Tissue Res. 2014;355(2):267-278.[67]Suadicani SO, Cherkas PS, Zuckerman J, et al. Bidirectional calcium signaling between satellite glial cells and neurons in cultured mouse trigeminal ganglia. Neuron Glia Biol. 2010; 6(1):43-51.[68]Warwick RA, Hanani M. Involvement of aberrant calcium signalling in herpetic neuralgia. Exp Neurol.2016; 277:10-18.[69]Chen X , Pang RP , Shen KF , et al. TNF-α enhances the currents of voltage gated sodium channels in uninjured dorsal root ganglion neurons following motor nerve injury. Exp Neurol.2011;227(2):279-286.[70]Zhang X, Liu C, Nestor KE , et al. The effect of glypican-1 glycosaminoglycan chains on turkey myogenic satellite cell proliferation, differentiation, and fibroblast growth factor 2 responsiveness.Poult Sci.2007; 86(9):2020-2028.[71]Vicente R, Escalada A, Soler C, et al. Pattern of Kv beta subunit expression in macrophages depends upon proliferation and the mode of activation. J Immunol. 2005; 174(8):4736.[72]Pang RP, Xie MX, Yang J , et al. Downregulation of ClC-3 in dorsal root ganglia neurons contributes to mechanical hypersensitivity following peripheral nerve injury. Neuropharmacology.2016;110:181-189.[73]Song DD , Li Y , Tang D , et al. Neuron-glial communication mediated by TNF-? and glial activation in dorsal root ganglia in visceral inflammatory hypersensitivity. Am J Physiol Gastrointest Liver Physiol. 2014;306(9):G788-795.[74]Qiao LN, Tan LH, Yang JJ, et al. Effects of Electroacupuncture on Activities of Satellite Glial Cells of Dorsal Root Ganglia in Rats with Neck Incision Pain.Zhen Ci Yan Jiu. 2017;42(4):283-289.[75]李瑞午,李翠红,汪智民. 足三里穴区不同层次感觉和运动神经元的节段性分布研究[J].中国针灸, 2000,20(3):161-163.[76]边冠鹤,李泽山.猫大肠各部的初级传入神经来源及节段性分布—HRP法研究[J]. 锦州医科大学学报,1993(2):1-6.[77]Tang Y, Yin HY, Liu J,et al. P2X receptors and acupuncture analgesia.Brain Res Bull. 2018;17:1-28. |
| [1] | Li Dadi, Zhu Liang, Zheng Li, Zhao Fengchao. Correlation of total knee arthroplasty efficacy with satisfaction and personality characteristics [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1346-1350. |
| [2] | Wei Wei, Li Jian, Huang Linhai, Lan Mindong, Lu Xianwei, Huang Shaodong. Factors affecting fall fear in the first movement of elderly patients after total knee or hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1351-1355. |
| [3] | Lü Zhen, Bai Jinzhu. A prospective study on the application of staged lumbar motion chain rehabilitation based on McKenzie’s technique after lumbar percutaneous transforaminal endoscopic discectomy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1398-1403. |
| [4] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Comparison of the short-term efficacy of extracorporeal shock wave therapy for middle-aged and elderly knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1471-1476. |
| [5] | Gao Yan, Zhao Licong, Zhao Hongzeng, Zhu Yuanyuan, Li Jie, Sang Deen. Alteration of low frequency fluctuation amplitude at brain-resting state in patients with chronic discogenic low back pain [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1160-1165. |
| [6] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [7] | Wang Feng, Zhou Liyu, Saijilafu, Qi Shibin, Ma Yanxia, Wei Shanwen. CaMKII-Smad1 promotes axonal regeneration of peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1064-1068. |
| [8] | Zhao Zhongyi, Li Yongzhen, Chen Feng, Ji Aiyu. Comparison of total knee arthroplasty and unicompartmental knee arthroplasty in treatment of traumatic osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 854-859. |
| [9] | Zhang Nianjun, Chen Ru. Analgesic effect of cocktail therapy combined with femoral nerve block on total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 866-872. |
| [10] | Wu Gang, Chen Jianwen, Wang Shilong, Duan Xiaoran, Liu Haijun, Dong Jianfeng. Simple HyProCure subtalar stabilization in treatment of adolescent flexible flatfoot combined with painful accessory navicular bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 901-905. |
| [11] | Li Yan, Wang Pei, Deng Donghuan, Yan Wei, Li Lei, Jiang Hongjiang. Electroacupuncture for pain control after total knee arthroplasty: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 957-963. |
| [12] | Li Quanxi, Shen Yu, Wan Wei, Sun Shanzhi. Changes of abdominal wall mechanics and pain after tension-free inguinal hernia repair with polypropylene mesh [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 548-552. |
| [13] | Wang Xiaofei, Teng Xueren, Cong Linyan, Zhou Xu, Ma Zhenhua. Herbert screw internal fixation for treating adult osteochondritis dissecans of the knees [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 397-402. |
| [14] | Yang Wei, Chen Zehua, Yi Zhiyong, Huang Xudong, Han Qingmin, Zhang Ronghua. Effectiveness of intra-articular injection of hyaluronic acid versus placebo in the treatment of early and mid-stage knee osteoarthritis: a Meta-analysis based on randomized, double-blind, controlled, clinical trials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3760-3766. |
| [15] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||