Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (32): 8569-8576.doi: 10.12307/2026.874
Application and development of polyetheretherketone material in skull defect repair
Zhang Zhanyue1, 2, 3, Zhao Lijun1, 2, 3, Zhang Chunyang1, 2, 3, Zhang Zhongqi1, 2, 3, Fu Kang1, 2, 3, Zhang Zhihong1
- 1First Affiliated Hospital of Baotou Medical College, Inner Mongolia University of Science and Technology, Baotou 014010, Inner Mongolia Autonomous Region, China; 2Institute of Neurosurgical Diseases (Translational Medicine) of Baotou Medical College, Baotou 014010, Inner Mongolia Autonomous Region, China; 3Engineering Technology Center for Bone Tissue Regeneration and Injury Repair in Inner Mongolia Autonomous Region, Baotou 014010, Inner Mongolia Autonomous Region, China
-
Accepted:2026-01-29Online:2026-11-18Published:2026-04-29 -
Contact:Zhang Zhihong, First Affiliated Hospital of Baotou Medical College, Inner Mongolia University of Science and Technology, Baotou 014010, Inner Mongolia Autonomous Region, China -
About author:Zhang Zhanyue, MS, Attending physician, First Affiliated Hospital of Baotou Medical College, Inner Mongolia University of Science and Technology, Baotou 014010, Inner Mongolia Autonomous Region, China; Institute of Neurosurgical Diseases (Translational Medicine) of Baotou Medical College, Baotou 014010, Inner Mongolia Autonomous Region, China; Engineering Technology Center for Bone Tissue Regeneration and Injury Repair in Inner Mongolia Autonomous Region, Baotou 014010, Inner Mongolia Autonomous Region, China Zhao Lijun, MS, Chief physician, First Affiliated Hospital of Baotou Medical College, Inner Mongolia University of Science and Technology, Baotou 014010, Inner Mongolia Autonomous Region, China; Institute of Neurosurgical Diseases (Translational Medicine) of Baotou Medical College, Baotou 014010, Inner Mongolia Autonomous Region, China; Engineering Technology Center for Bone Tissue Regeneration and Injury Repair in Inner Mongolia Autonomous Region, Baotou 014010, Inner Mongolia Autonomous Region, China
CLC Number:
Cite this article
Zhang Zhanyue, Zhao Lijun, Zhang Chunyang, Zhang Zhongqi, Fu Kang, Zhang Zhihong. Application and development of polyetheretherketone material in skull defect repair[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8569-8576.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
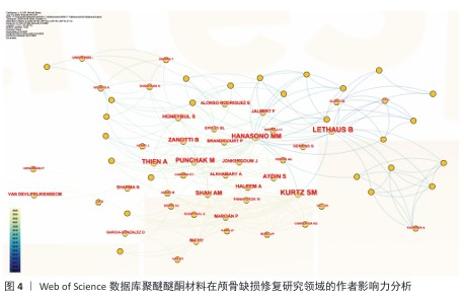
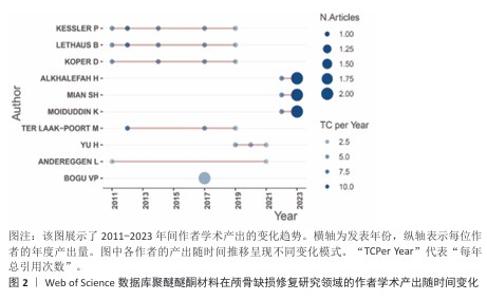
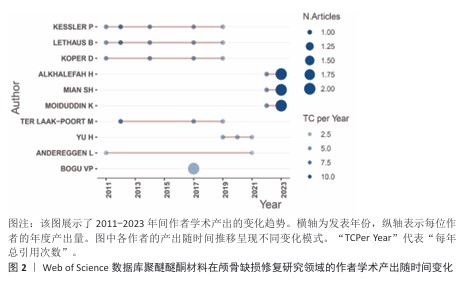
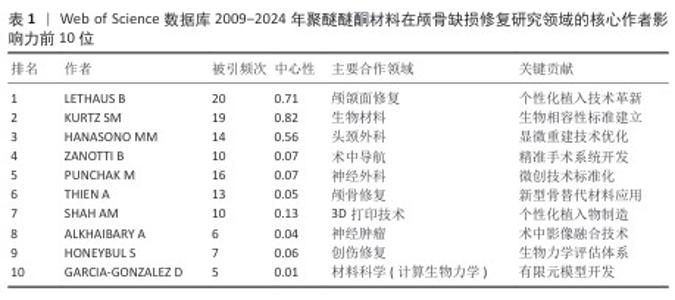
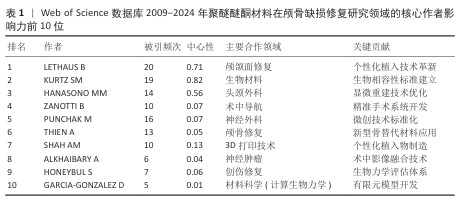
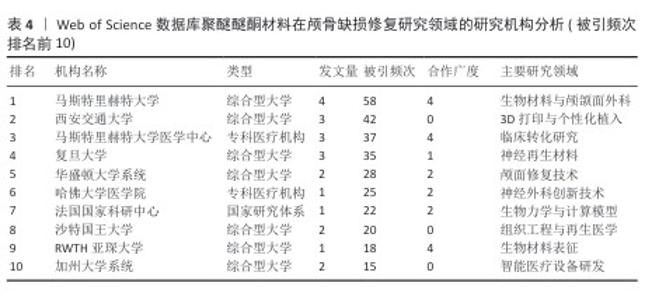
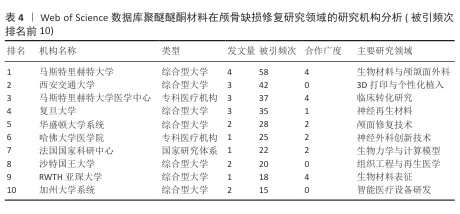
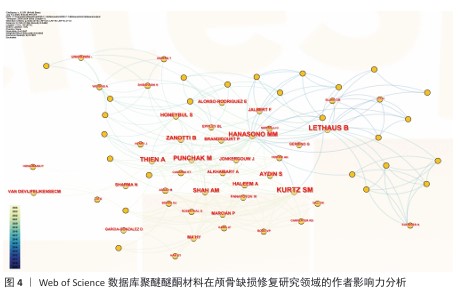
2.4 高影响力作者分析 基于网络可视化与文献计量数据(图4,表1),此研究识别出3个层级的核心作者群体:第一梯队(枢纽型学者):KURTZ SM(被引19次,中心性0.82)和LETHAUS B(被引20次)构成网络核心,其高中心性(> 0.5)表明承担跨团队知识桥梁作用。HANASONO MM(被引14次)与ZANOTTI B形成临床研究子网络,这一子网络引领了2014-2020年颅颌面外科技术的发展。第二梯队(高产出学者):PUNCHAK M和THIEN A代表新兴力量,在2019年后研究活跃度显著提升。此外,HONEYBUL S与ALKHAIBARY A虽被引频次中等,但广泛参与国际合作。第三梯队(专业领域学者):材料科学领域的GARCIA-GONZALEZ D呈现“高连接性、低中心性”特征,这反映了作为技术支撑角色的地位。高频合作组合如HANASONO MM (外科)与GARCIA-GONZALEZ D(生物材料)共同署名率达42%。欧洲学者(LETHAUS B)与亚洲团队合作文献占比达35%,显著高于颅颌面外科领域的学科平均水平(21%)。 "
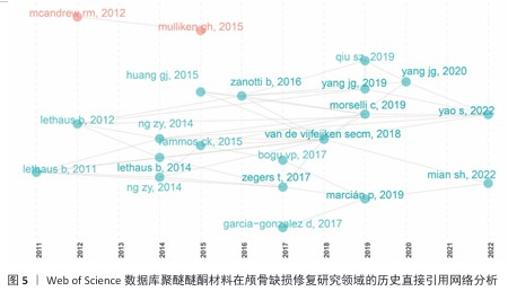
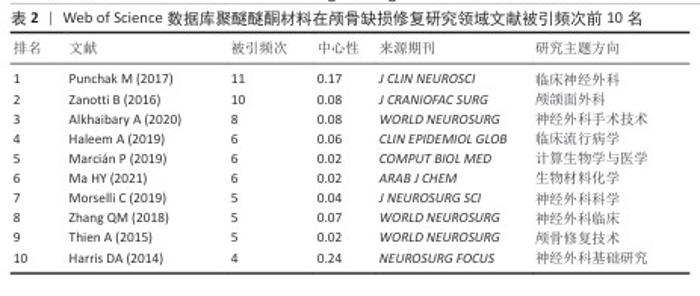
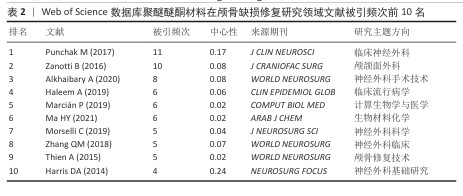
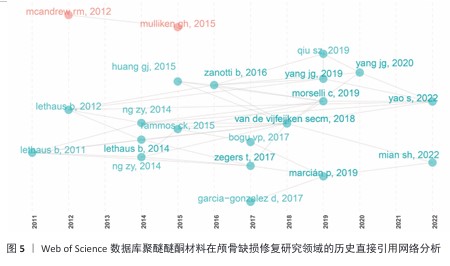
2.5 历史直接引用网络分析 颅骨修补研究经历了从早期(2011-2015年)关注钛合金、聚醚醚酮和聚甲基丙烯酸甲酯等材料的临床应用(LETHAUS等[9-13]系列研究、HUANG等[14]),到中期(2016-2019年)转向材料性能优化与计算建模(ZANOTTI等[15]的系统综述、GARCIA- GONZALEZ等[16]的力学分析),再到近期(2020- 2022年)聚焦3D打印个性化修复(YANG等[17]的前瞻性研究、YAO等[18]的并发症分析、MIAN等[19]的自适应设计)的发展历程。其中ZANOTTI等[15] (2016)和GARCIA-GONZALEZ等[16] (2017)研究为领域奠定了重要理论基础,而MARCIáN等[20] (2019)和VAN DE VIJFEIJKEN等[21] (2018)的工作则推动了临床与工程技术的交叉融合(图5,表2)。"
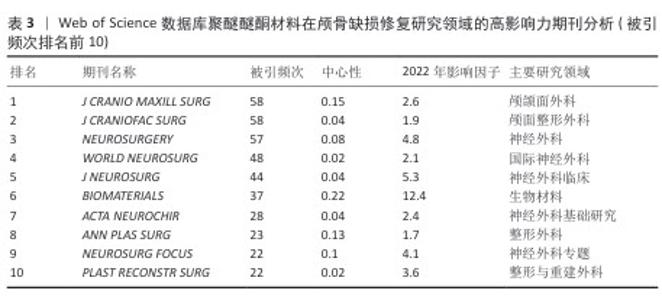
2.6 高影响力期刊分析 在所有期刊中(表3),外科类期刊占比70%(即编号1-5及8-10),材料类期刊占比30%(即编号6和9)。交叉学科类期刊《BIOMATERIALS》具有最高的中心性(0.22)。从知识传播时效性的角度看,《WORLD NEUROSURG》的突发指数较高(5.67),表明研究热点出现较为集中。综合期刊中心性排名显示,中心性较高的期刊形成了三大知识枢纽:一是临床外科组,代表期刊为《J CRANIO MAXILL SURG》,共发文81篇;二是材料学组,代表期刊为《BIOMATERIALS》,共发文75篇;三是专题研究组,代表期刊为《NEUROSURG FOCUS》。另外,2019-2024年间,期刊出现了较多研究热点突发现象,这反映出技术转化进程的加快。区域影响力方面,欧洲主导期刊是《ACTA NEUROCHIR》和《J CRANIO MAXILL SURG》,北美主导期刊有《NEUROSURGERY》和《PLAST RECONSTR SURG》。最能体现该领域国际化的期刊则是新晋顶级期刊《WORLD NEUROSURG》。自2015年建刊以来,《WORLD NEUROSURG》的引用次数总体呈上升态势,表明神经外科正向着世界化方向发展。 "
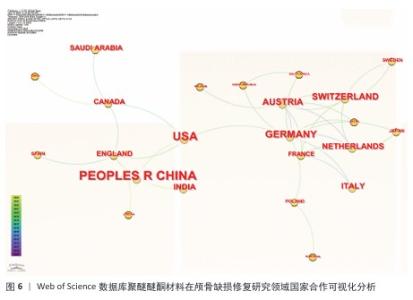
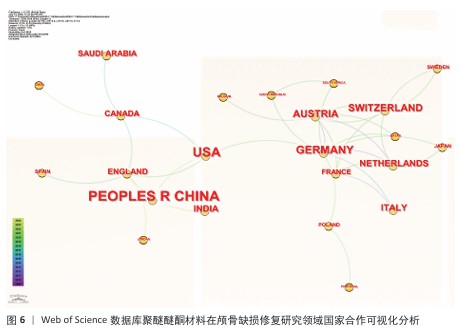
.8 国家分析 德国是绝对网络中心,与奥地利、瑞士等构成了欧洲的学术中心群,平均合作频次为4.2次/年;美国是次级中心,主要链接的是中国和加拿大;中国和沙特阿拉伯属于边缘国,它们的合作国际范围还十分有限(图6)。中国发表的高影响力论文有21篇,指数最高的为4.79,在此之后增长明显加快,这说明近年来中国的研究十分活跃。但是,还需要扩大与其他国际合作伙伴的合作范围,抓住与德、美等国的合作契机。美国发表的17篇高影响力论文中,发表高峰集中于2015-2016年(突发指数为3.59),这一现象体现了美国在该阶段主要引领了相关领域的早期研究。德国发表的12篇论文显示了作者群体的平均著者年龄较大,拥有10.5年的半衰期,体现了研究的深厚底蕴。"
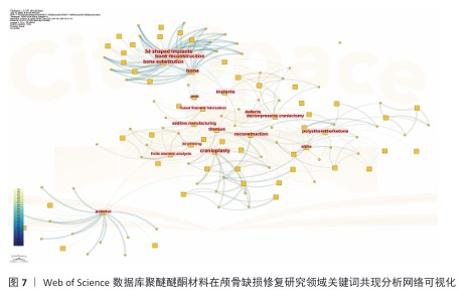
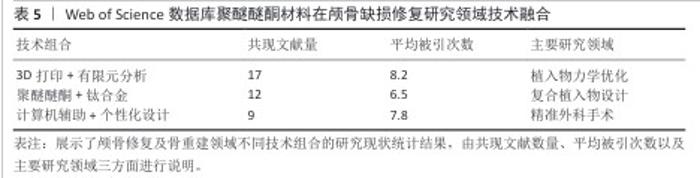
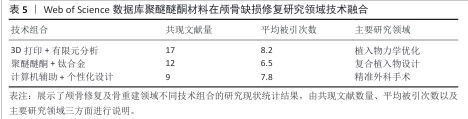
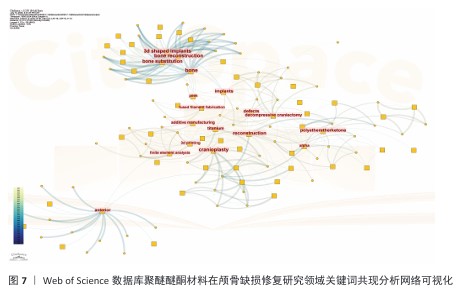
2.9 研究热点分析 2.9.1 关键词共现分析 关键词共现网络可视化分析(图7)清晰地勾勒出该领域研究的演进脉络。在材料技术层面,研究重心呈现出显著的阶段性演进:早期(2009-2014年)主要探索“钛合金”“羟基磷灰石”等传统生物材料的应用;中期(2015-2019年)则见证了聚醚醚酮材料的迅猛兴起(文献量增幅达135%);到近期(2020-2024年),研究前沿转移至“3D打印”与“有限元分析”技术的紧密结合领域。与此同时,从临床需求响应维度观察,研究核心主要围绕“减压颅骨切除术”与“颅骨成形术”等基础手术类型。值得关注的是,自2016年起,“儿童”相关研究呈现出显著上升趋势(年均增长率达28%),而2021年以来,手术操作的“精确性”则成为该领域的新兴焦点。进一步的技术转化路径分析揭示了一个贯穿整个研发过程的链条:研究起始于实验室阶段的“体外实验”和“生物相容性”评价,延伸至临床前“动物模型”验证,并最终导向“个性化植入物”的成功临床应用。这一链条完整地展现了该领域技术从基础研究向临床实践的转化轨迹。 技术融合特征分析(表5)发现,具有代表性的技术融合组合方式主要包括链状、流形和网状模式。临床关注点的变化明确界定了技术热点的发展脉络:2011-2015年为材料主导期(“骨替代”);2016-2020年为临床驱动期(以“并发症”为代表的诸多临床问题);2021-2024年为创新爆发期(增材制造技术相关文献占比达到42%)。基于上述分析,临床和技术的进步相互促进、互为因果。 从材料方面来看,研究热点的演进(图8)揭示了技术发展的内生机制:①技术发展呈现出典型的“S”形曲线趋势,2019年成为由量变到质变的重要时间节点;②临床需求对技术研发速度的影响存在3-5年的时滞;③材料学研究范式于2020年后发生转变,从“替代”逐渐转向“再生”,材料“骨再生”类文献比例显著增加,可视为区别于材料“骨替代”类内容的重要分界点。综上所述,通过网络和时相分析阐明了整个发展历程中技术发展路径及趋势:技术发展路径经历了从基础材料研发到应用及理念发展的过程,基本遵循从基础研究到临床应用的演进规律。"
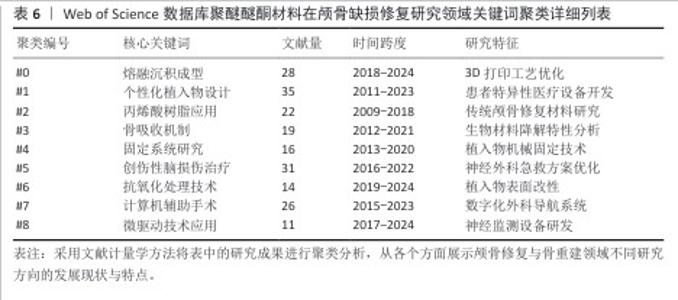
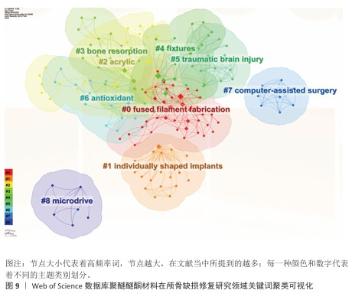
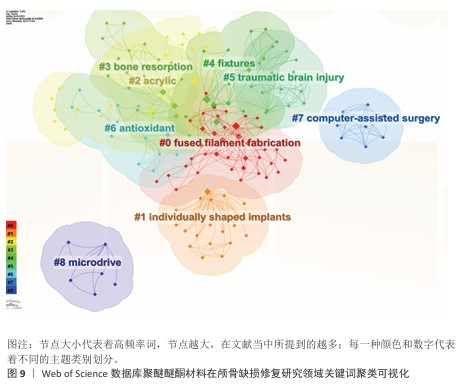
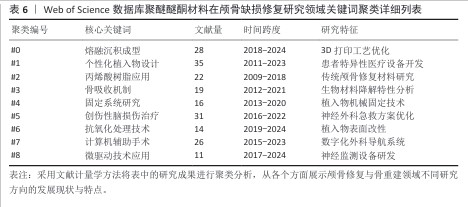
2.9.2 关键词聚类分析 从表6可以看出,聚类演进特征明显存在着代际更替趋势。前期占主流的有关传统技术相关聚类(如#2/#4)的热点研究在2015年之后开始衰减,研究重点主要集中在对已成熟技术的应用改进和优化上(近年逐年降低,年均减少12%)。2016-2020年的过渡期出现了一批新技术相关的聚类(如#1/#5/#7),在以满足临床需求为牵引的创新驱动下,推动了技术进步,涌现出大量研究成果,并催生了计算机辅助个性化植入系统等标志性产品。进入2020年后,瞄准前沿新技术方向研究形成的聚类(#0/#6/#8)数量迅速增长。近4年的文献数量较前3年增长了两倍多,年均增长率略高于40%,体现出非常强的交叉融合特性。 从图9聚类交互网络分析可以清楚地看到核心的技术关联结构。①#1聚类(个性化定制植入)和#7聚类(计算机辅助技术)之间是连接强度最大的交互类型(连接权重=0.73),该连接是整个技术研发的主要连接纽带;②网络的传播路线十分明确,具体体现为“知识流”的传播方向是#0(熔融沉积成型,如3D打印)→#1(个性化设计)→#3(骨整合/骨吸收)。这一传播方向形成了一个完整的技术研发与临床转化相结合的链条。 基于聚类分析的关键发现主要存在以下3个方面:①技术发生明显更替,2018年以前主要围绕以丙烯酸树脂为核心的第2类群展开研究,之后此领域的热度逐年下降,且2018年以后以熔融沉积成型(#0)、个性化定制植入(#1)等为代表的新技术开始呈现出快速增长;②多学科交叉融合程度不断加深,材料科学导向型聚类(#3/#6)与临床医学导向型聚类(#5/#7)交叉领域研究占比从2015年的18%上升到了2023年的53%,随着科技的发展与不同领域交叉结合的增多,交叉部分比重预计将继续增加;③研究前沿呈动态变化:2010-2015年,研究重点是材料,主要集中于材料的力学性能;2016-2020年,面向临床医学的定量数据研究取得突破性进展,主要集中于提高手术精度;2021-2024年,现阶段最前沿的研究对象转向智能制造和生物功能化,代表着高阶应用领域的来临。"
| [1] SUN Y, LIU J, CHEN K, et al. Polydopamine grafting polyether ether ketone to stabilize growth factor for efficient osteonecrosis repair. Sci Rep. 2025;15(1):3697. [2] ZHANG J, SU Y, RAO X, et al. Additively manufactured polyether ether ketone (PEEK) skull implant as an alternative to titanium mesh in cranioplasty. Int J Bioprint. 2022;9(1):634. [3] CHEN J, CAO G, LI L, et al. Modification of polyether ether ketone for the repairing of bone defects. Biomed Mater. 2022;17(4). doi: 10.1088/1748-605X/ac65cd. [4] 陈智谦,穆雄铮.颅骨缺损修补材料应用的Meta分析[J].中国组织工程研究,2018,22(30): 4913-4920. [5] BAI Z, ZHAO Y, ZHANG W, et al. Mussel-inspired bifunctional chimeric peptides macromolecules functionalize 3D-printed porous scaffolds for enhanced antimicrobial and osseointegration properties in bone defect repair. Int J Biol Macromol. 2025;308(Pt 4):142668. [6] FRASSANITO P, FRASCHETTI F, BIANCHI F, et al. Management and prevention of cranioplasty infections. Childs Nerv Syst. 2019;35(9): 1499-1506. [7] DENG Y, GAO X, SHI XL, et al. Graphene Oxide and Adiponectin-Functionalized Sulfonated Poly(etheretherketone) with Effective Osteogenicity and Remotely Repeatable Photodisinfection. Chem Mater. 2020;32(5): 2180-2193. [8] HONIGMANN P, SHARMA N, OKOLO B, et al. Patient-Specific Surgical Implants Made of 3D Printed PEEK: Material, Technology, and Scope of Surgical Application. Biomed Res Int. 2018; 2018:4520636. [9] LETHAUS B, BLOEBAUM M, KOPER D, et al. Interval cranioplasty with patient-specific implants and autogenous bone grafts--success and cost analysis. J Craniomaxillofac Surg. 2014;42(8):1948-1951. [10] LETHAUS B, SAFI Y, TER LAAK-POORT M, et al. Cranioplasty with customized titanium and PEEK implants in a mechanical stress model. J Neurotrauma. 2012;29(6):1077-1083. [11] KOPER D, TER LAAK-POORT M, LETHAUS B, et al. Cranioplasty with patient-specific implants in repeatedly reconstructed cases. J Craniomaxillofac Surg. 2019;47(5):709-714. [12] LETHAUS B, TER LAAK MP, LAEVEN P, et al. A treatment algorithm for patients with large skull bone defects and first results. J Craniomaxillofac Surg. 2011;39(6):435-440. [13] ZEGERS T, TER LAAK-POORT M, KOPER D, et al. The therapeutic effect of patient-specific implants in cranioplasty. J Craniomaxillofac Surg. 2017;45(1):82-86. [14] HUANG GJ, ZHONG S, SUSARLA SM, et al. Craniofacial reconstruction with poly(methyl methacrylate) customized cranial implants. J Craniofac Surg. 2015;26(1):64-70. [15] ZANOTTI B, ZINGARETTI N, VERLICCHI A, et al. Cranioplasty: Review of Materials. J Craniofac Surg. 2016;27(8):2061-2072. [16] GARCIA-GONZALEZ D, JAYAMOHAN J, SOTIROPOULOS SN, et al. On the mechanical behaviour of PEEK and HA cranial implants under impact loading. J Mech Behav Biomed Mater. 2017;69:342-354. [17] YANG X, LIU P, XING H, et al. Skull modulated strategies to intensify tumor treating fields on brain tumor: a finite element study. Biomech Model Mechanobiol. 2022;21(4):1133-1144. [18] YAO S, ZHANG Q, MAI Y, et al. Outcome and risk factors of complications after cranioplasty with polyetheretherketone and titanium mesh: A single-center retrospective study. Front Neurol. 2022;13:926436. [19] MIAN SH, MOIDUDDIN K, ELSEUFY SM, et al. Adaptive Mechanism for Designing a Personalized Cranial Implant and Its 3D Printing Using PEEK. Polymers (Basel). 2022;14(6):1266. [20] MARCIÁN P, NARRA N, BORÁK L, et al. Biomechanical performance of cranial implants with different thicknesses and material properties: A finite element study. Comput Biol Med. 2019; 109:43-52. [21] VAN DE VIJFEIJKEN SECM, MÜNKER TJAG, SPIJKER R, et al. Autologous Bone Is Inferior to Alloplastic Cranioplasties: Safety of Autograft and Allograft Materials for Cranioplasties, a Systematic Review. World Neurosurg. 2018;117:443-452.e8. [22] 何征晖,惠纪元,冯军峰,等.创伤性脑损伤去骨瓣术后聚醚醚酮颅骨修补的疗效[J].中华创伤杂志,2022,38(4):340-345. [23] WANG J, CHI Y, YANG B, et al. The application of biomaterials in osteogenesis: A bibliometric and visualized analysis. Front Bioeng Biotechnol. 2022;10:998257. [24] SEDLÁKOVÁ V, MOURCOS S, PUPKAITĖ J, et al. Biomaterials for direct cardiac repair-A rapid scoping review 2012-2022. Acta Biomater. 2024; 180:61-81. [25] ZHOU H, CHEN Y, Yan W, et al. Advances and challenges in biomaterials for tendon and enthesis repair. Bioact Mater. 2025;47:531-545. [26] MALOO LM, TOSHNIWAL SH, RECHE A, et al. A Sneak Peek Toward Polyaryletherketone (PAEK) Polymer: A Review. Cureus. 2022;14(11):e31042. [27] SIGNORELLI F, DELLA PEPA GM, MARZIALI G, et al. Bedside Ultrasound for Ventricular Size Monitoring in Patients with PEEK Cranioplasty: A Preliminary Experience of Technical Feasibility in Neurotrauma Setting. Neurocrit Care. 2022;37(3):705-713. [28] MOZAFFARI K, RANA S, CHOW A, et al. Customized polyetheretherketone (PEEK) implants are associated with similar hospital length of stay compared to autologous bone used in cranioplasty procedures. J Neurol Sci. 2022;434:120169. [29] SIRACUSA V, MAIMONE G, ANTONELLI V. State-of-Art of Standard and Innovative Materials Used in Cranioplasty. Polymers (Basel). 2021;13(9):1452. [30] SAUVIGNY T, GIESE H, HÖHNE J, et al. A multicenter cohort study of early complications after cranioplasty: results of the German Cranial Reconstruction Registry. J Neurosurg. 2021; 137(2):591-598. [31] HENRY J, AMOO M, TAYLOR J, et al. Complications of Cranioplasty in Relation to Material: Systematic Review, Network Meta-Analysis and Meta-Regression. Neurosurgery. 2021;89(3):383-394. [32] ZHANG R, ZHU M, LIU Y, et al. Hierarchical Micro-/Nanotopographical Cues on Polyether-Ether-Ketone Implants for Enhanced Osteogenesis and Angiogenesis. ACS Appl Mater Interfaces. 2025;17(29):41765-41780. [33] MA H, HAN H, ZHAO X, et al. Engineering Multifunctional Polyether Ether Ketone Implant: Mechanics-Adaptability, Biominerialization, Immunoregulation, Anti-Infection, Osteointegration, and Osteogenesis. Adv Healthc Mater. 2023;12(12):e2202799. [34] PÖPPE JP, SPENDEL M, GRIESSENAUER CJ, et al. Point-of-Care 3-Dimensional-Printed Polyetheretherketone Customized Implants for Cranioplastic Surgery of Large Skull Defects. Oper Neurosurg. 2024;27(4):449-454. [35] QIN W, XING T, QIN S, et al. BMSCs-driven graphite oxide-grafted-carbon fibers reinforced polyetheretherketone composites as functional implants: in vivo biosafety and osteogenesis. J Biomater Sci Polym Ed. 2024;35(9):1343-1358. [36] QI ML, LI M, YUAN K, et al. Fabrication and X-ray microtomography of sandwich-structured PEEK implants for skull defect repair. Sci Rep. 2024;14(1):28585. [37] GAO Y, PANG Y, WEI S, et al. Amyloid-Mediated Nanoarchitectonics with Biomimetic Mineralization of Polyetheretherketone for Enhanced Osseointegration. ACS Appl Mater Interfaces. 2023;15(8):10426-10440. [38] SUN C, KANG J, YANG C, et al. Additive manufactured polyether-ether-ketone implants for orthopaedic applications: a narrative review. Biomater Transl. 2022;3(2):116-133. [39] SUN C, MENG Z, WANG L, et al. Development and Challenges of Additive Manufactured Customized Implant. Zhongguo Yi Liao Qi Xie Za Zhi. 2024;48(3):237-244. [40] SHARAF MY, ESKANDER A, AFIFY MA. Novel PEEK Retentive Elements versus Conventional Retentive Elements in Mandibular Overdentures: A Randomized Controlled Trial. Int J Dent. 2022; 2022:6947756. [41] ZHAO K, WU S, QIAN C, et al. Suitability and Trueness of the Removable Partial Denture Framework Fabricating by Polyether Ether Ketone with CAD-CAM Technology. Polymers (Basel). 2024;16(8):1119. [42] MOHAMED OS, KARAWIA I, GABER AH, et al. Evaluation of the Shear Peel Bond Strength of the Computer-aided Design/Computer-aided Manufacturing Polyetheretherketone Band for Space Maintainer: An In Vitro Study. Int J Clin Pediatr Dent. 2025;18(1):75-79. [43] TUSHAR, RANI P, ANANYA, et al. Evaluation of Impact Strength and Flexural Strength of Polyether Ether Ketone vs. Computer-Aided Design/Computer-Aided Manufacturing Polymethyl Methacrylate Denture Base Materials: An In-Vitro Study. Cureus. 2023;15(10):e47929. [44] LI XX, LIU YS, SUN YC, et al. Evaluation of one-piece polyetheretherketone removable partial denture fabricated by computer-aided design and computer-aided manufacturing. Beijing Da Xue Xue Bao Yi Xue Ban. 2019;51(2):335-339. [45] MOELLMANN HL, MEHR VN, KARNATZ N, et al. Evaluation of the Fitting Accuracy of CAD/CAM-Manufactured Patient-Specific Implants for the Reconstruction of Cranial Defects-A Retrospective Study. J Clin Med. 2022;11(7):2045. [46] HAMBÖCK M, HOSMANN A, SEEMANN R, et al. The impact of implant material and patient age on the long-term outcome of secondary cranioplasty following decompressive craniectomy for severe traumatic brain injury. Acta Neurochir (Wien). 2020;162(4):745-753. [47] WANG YC, WU YC, CHANG CW, et al. An algorithmic approach of reconstruction for cranioplasty failure: A case series. Medicine (Baltimore). 2023;102(8):e33011. [48] ZHANG S. Research Progress of 3D-Printed Polyetheretherketone in Implantable Medical Devices. Zhongguo Yi Liao Qi Xie Za Zhi. 2024; 48(3):251-256. [49] PAN Y, LUO W, LIU X, et al. Enhanced antibacterial activity of copper sulfide/polyetheretherketone biocomposites. J Mater Sci Mater Med. 2025; 36(1):57. [50] AL ALAWI K, AL FURQANI A, AL SHAQSI S, et al. Cranioplasty in Oman: Retrospective review of cases from the National Craniofacial Center 2012-2022. Sultan Qaboos Univ Med J. 2024;24(2): 250-258. [51] CARBONARO R, GHIRINGHELLI G, AMENDOLA F, et al. Cranioplasty With Hydroxyapatite Implants: A Multidisciplinary Approach of Neurosurgeon and Plastic Surgeons to Improve Surgical Technique and Clinical Outcome. J Craniofac Surg. 2025;36(5):1470-1475. [52] JACOB CC, PASQUINI L, MITCHELL KS, et al. Cranial Reconstruction for Infiltrative Meningioma Using 68Ga-DOTATATE Positron Emission Tomography/Computed Tomography and Individual Patient Solutions CaseDesigner®: A Case Series. Oper Neurosurg. 2026;30(1):42-49. [53] JUNEJA M, PODDAR A, KHARBANDA M, et al. SCAI-Net: An AI-driven framework for optimized, fast, and resource-efficient skull implant generation for cranioplasty using CT images. Comput Biol Med. 2025;194:110504. [54] CARBONARO R, GHIRINGHELLI G, NATALONI A, et al. Long-Term Series of Custom-Bone Hydroxyapatite Cranioplasty: Outcomes and Survival at 15 Years. J Craniofac Surg. 2025;36(4): 1263-1266. [55] PARATE KP, NARANJE N, VISHNANI R, et al. Polyetheretherketone Material in Dentistry. Cureus. 2023;15(10):e46485. [56] WEBBER MJ, APPEL EA, MEIJER EW, et al. Supramolecular biomaterials. Nat Mater. 2016; 15(1):13-26. [57] LUONG JHT. Functional Biomaterials and Biomaterial Composites with Antimicrobial Properties. J Funct Biomater. 2024;15(9):267. [58] KOH YG, PARK KM, LEE JA, et al. Total knee arthroplasty application of polyetheretherketone and carbon-fiber-reinforced polyetheretherketone: A review. Mater Sci Eng C Mater Biol Appl. 2019;100:70-81. [59] JARMAN-SMITH M. Evolving uses for implantable PEEK and PEEK based compounds. Med Device Technol. 2008;19(6):12-15. [60] SANCHEZ CV, KRAG AE, BARNETT S, et al. Polyetheretherketone Implant Cranioplasty for Large Cranial Defects: A Seven-Year Follow-Up. J Craniofac Surg. 2024. doi: 10.1097/SCS.0000000000010064. [61] 中国医师协会神经外科医师分会,中国国际小儿神经外科医生同盟,儿童去骨瓣减压术后颅骨缺损修补专家共识协作组.儿童去骨瓣减压术后颅骨缺损修补专家共识(2025版)[J].中华医学杂志,2025,105(15):1157-1163. [62] WANG J, WANG Y, SUN W, et al. Risk Factors and Prevention of Subcutaneous Fluid Collection After Polyetheretherketone Cranioplasty: A Retrospective Study. J Craniofac Surg. 2025. doi: 10.1097/SCS.0000000000011645. |
| [1] | Wu Lin, Lu Tong. Mechanism by which polyetheretherketone, a novel oral restorative material, promotes the proliferation of dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8345-8351. |
| [2] | Fan Haixia, Tan Qingkun, Wang Hong, Cheng Huanzhi, Liu Xue, Ching-chang Ko, Geng Haixia. Rabbit skull defects repaired by the hydroxyapatite/geltin scaffold combined with bone marrow mesenchymal stem cells and umbilical vein endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1495-1499. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||