Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (47): 8248-8254.doi: 10.3969/j.issn.2095-4344.2013.47.017
Previous Articles Next Articles
Silicon nanowire biosensor in detecting tumor markers
Li Zeng-yao, Wang Tong
- Department of Endoscopy Surgery, Wuxi People’s Hospital Affiliated to Nanjing Medical University, Wuxi 214000, Jiangsu Province, China
-
Revised:2013-09-06Online:2013-11-19Published:2013-11-19 -
Contact:Wang Tong, Chief physician, Professor, Doctoral supervisor, Department of Endoscopy Surgery, Wuxi People’s Hospital Affiliated to Nanjing Medical University, Wuxi 214000, Jiangsu Province, China aanti@163.com -
About author:Li Zeng-yao★, Master, Department of Endoscopy Surgery, Wuxi People’s Hospital Affiliated to Nanjing Medical University, Wuxi 214000, Jiangsu Province, China daniu810@126.com -
Supported by:the National Natural Science Foundation of China, No. 81371683/H1819*
CLC Number:
Cite this article
Li Zeng-yao, Wang Tong. Silicon nanowire biosensor in detecting tumor markers[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(47): 8248-8254.
share this article

2.1 肿瘤标记物 肿瘤标记物是指由肿瘤细胞本身或人体内其他细胞对于肿瘤和其他良性病变发生应答而产生的含量明显高于正常值的一类物质。肿瘤标记物一般可在肿瘤患者的体液或病变组织中检测到,其多为蛋白质、多肽、激素和酶等。大多数恶性肿瘤早期并无明显症状,影像学、病理学检测等亦无法做出明确的早期诊断。而灵敏肿瘤标记物的检测对于肿瘤早期发现,治疗后评价,检测复发和转移等方面都具有一定的应用价值,可为患者争取治疗时间,延长患者生命[13]。目前,临床上已确认将甲胎蛋白、癌胚抗原、前列腺特异抗原、糖类抗原等列为肝癌、结直肠癌、前列腺癌、卵巢癌、胆管癌等恶性肿瘤的诊断依据。 需要指出的是,传统检测手段对于上述肿瘤标记物的检测一般只达到ng/mL水平,而已报道的硅纳米线生物传感器对肿瘤标记物的检测最低可达到fg/mL水平,为肿瘤标记物的临床检测提供了新的途径[14]。 2.2 硅纳米线生物传感器及其检测原理 硅纳米线是一种半导体构筑块,见图1所示[15-17],其电学性能优秀、尺寸小、比表面积较大,这使得硅纳米线非常适合于制造超高敏感性生物传感器。"
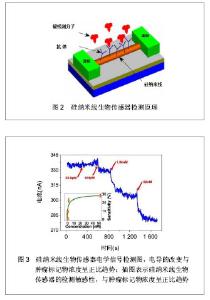
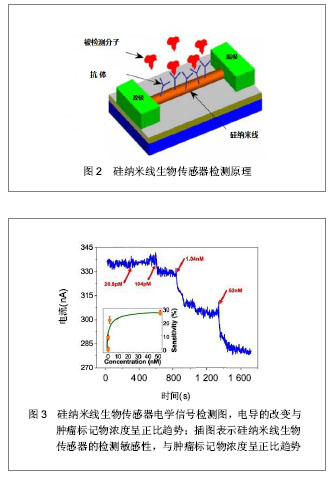
目前,硅纳米线的制备主要有2种方式:“自上而下”法和“自下而上”法[18-19]。前者是对晶体进行刻蚀、腐蚀或研磨,通过微纳加工技术获得硅纳米线[20];后者则主要通过化学气相沉积法将晶体制备成一定长度和宽度的硅纳米线[21]。基于硅纳米线的生物传感器是典型的以场效应晶体管为基础的器件,其具有源极、漏极和栅极。硅纳米线生物传感器的感应机制是:以硅纳米线半导体中电导的改变为基础,源极和漏极与半导体通路桥接,栅极对半导体的电导进行调节。其检测原理是:检测前,在硅纳米线表面修饰被检测目标分子的针对性受体。检测开始后,将交流电压或直流电压作用于硅纳米线传感器,其源极和漏极之间的半导体通路则成为器件的感应元件,带电荷的被检测分子直接与受体分子结合绑定后可引起感应元件电导的改变,见图2所示。因此,凭借被检测分子在硅纳米线生物传感器表面绑定后转变成的电学信号,最终可得到其相应的检测限[22-25],见图3所示。"
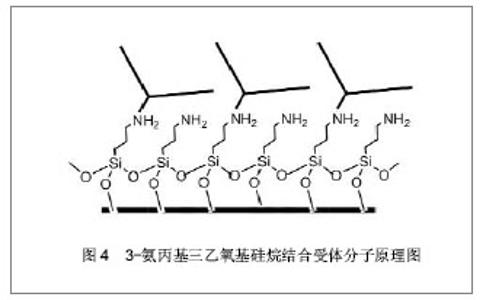
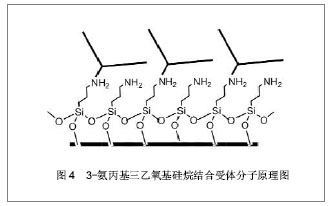
2.3 硅纳米线生物传感器的表面修饰 硅纳米线生物传感器在进行检测前需对硅纳米线表面进行特异性受体分子的修饰,以便识别特定的肿瘤标记物。目前最常用的表面修饰方法为共价结合法。共价结合是在不影响受体活性的前提下,通过某种连接分子将受体分子与硅纳米线表面共价连接。由于硅纳米线表面可被氧化,故依靠其表面的氧化层与某种化学试剂形成连接链即可完成。 目前已报道的可形成连接链的化学试剂有3-氨丙基三乙氧基硅烷、N-羟基琥珀酰亚胺和1- (3-二甲氨基丙基)-3-乙基碳二亚胺盐酸盐、3-氨丙基甲基二乙氧基硅烷、γ-甲基丙烯酰氧基丙基三甲氧基硅烷及3-氨基丙基-三甲氧基硅烷等[22-24, 26-27]。其中较为常用是3-氨丙基三乙氧基硅烷与受体分子共价连接后固定到硅纳米线表面[28],见图4所示。3-氨丙基三乙氧基硅烷含有的氨基团可以固定肽核酸、DNA和抗体等多种受体分子[29-35]。 此外,静电吸附作用也用于硅纳米线的表面修饰,其主要依靠的是溶质离子与带有相反电荷吸附剂的吸引作用。2006年,Bunimovich等[36]报道了一篇应用静电吸附作用对硅纳米线生物传感器表面成功修饰的研究。令人欣喜的是,2007年,Park等[37]又报道了1篇应用电阻加热法对硅纳米线表面进行选择性修饰,极大地促进了硅纳米线传感器表面修饰手段的发展。 "

2.5 德拜长度的影响 德拜长度是指带电荷的被检测分子电场的作用距离,超过这个距离,其作用基本上就被周围带有相反电荷离子的电场所屏蔽,这种电荷屏蔽作用就是德拜长度的主要特点。硅纳米线传感器对被检测分子电荷的检测与其德拜长度直接相关,也就是说,较长的德拜长度可以减弱硅纳米线表面的电荷屏蔽作用。 德拜长度可用公式λD=√εkBT/q2c推算出来,其中λD代表德拜长度,ε代表介质的介电常数,kB是波尔兹曼常数,T代表温度,q代表电荷数,而c为电解质溶液的离子浓度。由公式可知,在高离子浓度的溶液中,电荷屏蔽作用可从根本上阻碍电荷的检 测[40-43]。比如,在一个100 mmol/L的缓冲溶液中,德拜长度约为1 nm,所以被检测分子的表面电势在超过1 nm的范围内将会被完全屏蔽。因为,人体体液如血清的离子浓度也在100 mmol/L左右,因此,大多数硅纳米线生物传感器的检测在配置的标准品液中进行,应用的缓冲液离子浓度一般低于 10 mmol/L[44]。 2.6 硅纳米线生物传感器对肿瘤标记物的检测实例 目前,一些研究团队已经应用硅纳米线生物传感器成功完成了数种肿瘤标记物的检测,检测结果十分理想,展现了硅纳米线生物传感器良好的应用前景。然而由于德拜长度等因素的影响,目前大多数应用只能留在对标准品溶液的检测,对临床肿瘤患者血清的检测数据仍然缺乏。"


2.6.1 前列腺癌肿瘤标记物的检测 作为前列腺癌最有价值的肿瘤标记物,前列腺特异性抗原已被广泛应用于前列腺癌的筛选、早期诊断和分期上,其特异性高达97%[47-48]。2007年,Kim等[23]报道了应用n型硅纳米线生物传感器在微流体通路下对前列腺特异性抗原进行的超敏感检测。前列腺特异性抗原抗体应用3-氨丙基三乙氧基硅烷分子作为连接分子修饰到硅纳米线表面,在无需对抗原前列腺特异性抗原标记的情况下,对预先配置的不同浓度前列腺特异性抗原的标准品溶液进行检测,最终得到了前列腺特异性抗原超敏检测限<1 fg/mL。此外,作者同时指出控制硅纳米线尺寸和制作时的掺杂程度可以影响硅纳米线生物传感器的检测敏感性。遗憾的是,作者只进行了标准品溶液的检测,并没有对临床上前列腺癌患者的血清进行前列腺特异性抗原检测。 2.6.2 乳腺癌肿瘤标记物检测 糖类抗原CA15-3是乳腺癌的肿瘤标记物,30%-50%的患者其指标会明显升高,CA15-3也是乳癌术后监测的最佳指标[49-50]。Stern等[26]报道了通过射流净化芯片系统和硅纳米线生物传感器的整合,应用开放性液池对全血中CA15-3进行检测。射流净化芯片系统应用抗原抗体结合原理使肿瘤标记物从全血中分离,再通过光化学裂解得到纯化的肿瘤标记物,最后把纯化的肿瘤标记物转移到硅纳米线生物传感器中进行检测。Stern等获得CA15-3在全血中的检测值为30 U/mL。此检测方法能够克服硅纳米线生物传感器检测血清样本时非肿瘤标记物分子的非特异性绑定作用,增加传感器应用的临床相关性。但其纯化过程较为繁琐,相当于对肿瘤标记物进行了一次标记。 2.6.3 结直肠癌肿瘤标记物检测 癌胚抗原为结直肠癌的辅助诊断指标,其为正常胚胎组织所产生的成分,出生后逐渐消失,或仅存极微量。当细胞癌变时,此类抗原表达可明显增多。Zheng等[22]报道了应用硅纳米线生物传感器阵列在微流体通路下对癌胚抗原、前列腺特异性抗原、mucin-1三种肿瘤标记物配置液进行的同时多路复用检测。凭借该更为敏感的检测方法,Zheng等得到癌胚抗原的检测限为0.9 pg/mL。然而作者同样没有在血清样本中完成对癌胚抗原的检测。 2.6.4 原发性肝癌的肿瘤标记物检测 甲胎蛋白为一种糖蛋白,正常情况下,这种蛋白主要来自胚胎的肝细胞,胎儿出生约两周后甲胎蛋白从血液中消失,是诊断原发性肝癌的特异性肿瘤标记物,具有确立诊断、早期诊断、鉴别诊断的作用[51]。Ivanov等[46]报道了应用n型硅纳米线生物传感器在开放性液池下对甲胎蛋白标准品溶液进行检测,得到了10- 15 mol/L的检测限。然而,作者同样没有进行血清样本的检测。 2.6.5 其他肿瘤标记物的检测 血管内皮生长因子属血小板源性生长因子家族的生长因子,其能增加血管通透性,并具有血管生成功能[53]。恶性肿瘤的生长代谢、浸润转移和复发均与肿瘤的血供密切相关。因此,血管内皮生长因子作为一种广谱的肿瘤标记物对多种肿瘤的辅助诊断均有重要的临床价值,且对初诊肿瘤患者,血管内皮生长因子也是一个良好的检测指标。人类C-反应蛋白是感染和组织损伤时血浆浓度快速,急剧升高的主要急性期蛋白,恶性肿瘤患者C-反应蛋白大都升高,如C-反应蛋白与甲胎蛋白的联合检测,可用于肝癌与肝脏良性疾病的鉴别诊断,并且,C-反应蛋白测定用于肿瘤的治疗和预后都有积极意义。Lee等[24]报道了应用n型和p型硅纳米线生物传感器在开放性液池下分别对血管内皮生长因子标准品液进行检测,其得到的检测限为n型1.04 nmol/L,p型104 pmol/L。而Jung等[17]报到的应用p型硅纳米线生物传感器在微流体通路下对C-反应蛋白标准品液的检测,得到C-反应蛋白的超敏感检测限为1 fmol/L。同样遗憾的是这两次检测都没有在血清样本下进行。"

| [1] Meng J,Meng J,Duan J,et al.Carbon nanotubes conjugated to tumor lysate protein enhance the efficacy of an antitumor immunotherapy.Small.2008;4(9):1364-1370.
[2] Sato K,Hibara AM,Hisamoto H,et al.Microchip-based chemical and biochemical analysis systems.Adv Drug Deliv Rev.2003;55(3):379-391.
[3] Lai S,Wang S,Luo J,et al.Design of a compact disc like microfluidic platform for enzyme- link immunosorbent assay.Anal Chem.2004;76(7):1832-1837.
[4] Zhang S,Li X,Zhang F.CE-based simultaneous liquid-phase noncompetitive enzyme immunoassay for three tumor markers in human serum using electrochemical detection. Electrophoresis.2007;28(23):4427-4434.
[5] Al-Jamal WT,Al-Jamal KT,Bomans PH,et al. Functionalized-quantum-dot-liposome hybrids as multimodal nanoparticles for cancer.Small.2008;4:1406-1415.
[6] Fu Z,Yan F,Liu H,et al.A channel-resolved approach coupled with magnet-captured technique for multianalytechemiluminescent immunoassay.Biosens Bioelectron.2008;23(10):1422-1428.
[7] Anderson NL,Anderson NG.The human plasma proteome:history, character, and diagnostic prospects.Mol Cell Proteomics.2002;1:845-867.
[8] Qian WJ,Jacobs JM,Liu T,et al.Advances and challenges in liquid chromatography–mass spectrometry-based proteomics profiling for clinical applications.Mol Cell Proteomics.2006; 5(10):1727-1744.
[9] Huang KJ,Niu DJ,Xie WZ,et al.A disposable electrochemical immunosensor for carcinoembryonic antigen based on nano-Au/multi-walled carbon nanotubes-chitosans nanocomposite film modified glassy carbon electrode. Anal Chim Acta.2010;659:102-108.
[10] Liu J,Lau SK,Varma VA,et al.Multiplexed detection and characterization of rare tumor cells in Hodgkin's lymphoma with multicolor quantum dots.Anal Chem.2010;82:6237-6243.
[11] Liu G,Lin Y.Nanomaterial labels in electrochemical immunosensors and immunoassays.Talanta. 2007;74: 308-317.
[12] Lee MH,Lee DH,Jung SW,et al.Measurements of serum C-reactive protein levels in patients with gastric cancer and quantification using silicon nanowire arrays.Nanomedicine. 2010;6:78-83.
[13] Lu M,Whitelegge JP,Whelan SA,et a1.Hydrophobic fraetionation enhances novel protein detection by mass spectrometry in triple negative breast cancer.Proteomics Bioinform.2010;3(2):1-10.
[14] Kim A,Ah CS,Yu HY,et al.Ultrasensitive, label-free, and real-time immunodetection using silicon field-effect transistors.Appl Phys Lett.2007;91:103901.
[15] Cui Y,Wei Q,Park H,et al.Nanowire nanosensors for highly sensitive and selective detection of biological and chemical species.Science.2001;293:1289-1292.
[16] Patolsky F,Zheng G,Lieber CM.Fabrication of silicon nanowire devices for ultrasensitive, label-free, real-time detection of biological and chemical species.Nat Protoc.2006;1: 1711-1724.
[17] Lee MH,Lee KN,Jung WW,et al.Quantitative measurements of C-reactive protein using silicon nanowire arrays.Int J Nanomedicine.2008;3(1):117-124.
[18] Cui Y,Wei QQ,Park HK,et al.Nanowire nanosensors for highly sensitive and selective detection of biological and chemical species. Science.2001;293:1289-1292.
[19] Stern E,Klemic JF,Routenberg DA,et al.Label-Free Immunodetection with CMOS-Compatible Semiconducting Nanowires.Nature.2007;445:519-522.
[20] Zeng XB,Xu YY,Zhang SB,et al.Silicon nanowires grown on a pre-annealed Si substrate.J Cryst Growth.2003;247(1):13-16.
[21] Ciucci S,Angelo FD,Diligenti A,et al.Silicon nanowires fabricated by means of underetching technique.Microelectron Eng.2005;78-79:338-342.
[22] Zheng G,Patolsky F,Cui Y,et al.Multiplexed electrical detection of cancer markers with nanowire sensor arrays.Nat Biotechnol. 2005; 23(10):1294-1301.
[23] Kim A,Ah CS,Yu HY,et al.Ultrasensitive, label-free, and real-time immunodetection using silicon field-effect transistors.Appl Phys Lett. 2007;91:103901.
[24] Lee HS,Kim KS,Kim CJ,et al.Electrical detection of VEGFs for cancer diagnoses using anti-vascular endotherial growth factor aptamer-modified Si nanowire FETs.Biosens Bioelectron. 2009;24(6):1801-1805.
[25] Nair PR,Alam MA.Design considerations of silicon nanowire biosensors.IEEE Trans Electron Devices.2007;54(12): 3400,3408.
[26] Stern E,Vacic A,Rajan NK,et al.Label-free biomarker detection from whole blood.Nat Nanotechnol.2010;5(2): 138-142.
[27] Li Z,Chen Y,Li X,et al.Sequence-Specific Label-Free DNA. Sensors Based on Silicon Nanowires.Nano Lett.2004;4: 245-247.
[28] Thakurta SG,Subramanian A.Fabrication of dense, uniform aminosilane monolayers: A platform for protein or ligand immobilization.Colloids Surf A: Physicochem Eng Aspects. 2012;414(20):384-392.
[29] Zhang GJ,Luo ZH,Huang MJ,et al.An integrated chip for rapid, sensitive, and multiplexed detection of cardiac biomarkers from fingerprick blood.Biosens Bioelectron.2011;28(1): 459-463.
[30] Zhang GJ,Huang MJ,Luo ZH,et al.Highly sensitive and reversible silicon nanowire biosensor to study nuclear hormone receptor protein and response element DNA interactions.Thomsen Biosens Bioelectron.2010;26(2): 365-370.
[31] Zhang GJ,Chua JH,Chee RE,et al.Label-free direct detection of MiRNAs with silicon nanowire biosensors.Biosens Bioelectron. 2009;24:2504-2508.
[32] Zhang GJ,Luo ZHH,Huang MJ,et al.Morpholino- functionalized silicon nanowire biosensor for sequence-specific label-free detection of DNA.Biosens Bioelectron.2010;25(11):2447-2453.
[33] 33Zhang GJ,Zhang L,Huang MJ,et al.Silicon nanowire biosensor for highly sensitive and rapid detection ofDengue virus.Sens Actuator B: Chem.2010;146:138-144.
[34] Chua J,Chee RE,Agarwal A,et al.Label-free electrical detection of cardiac biomarker with complementary metal-oxide semiconductor-compatible silicon nanowire sensor arrays.Anal Chem.2009;81(15):6266-6271.
[35] Zhang GJ,Huang M,Ang J,et al.Self-assembled monolayer-assisted silicon nanowire biosensor for detection of protein-DNA interactions in nuclear extracts from breast cancer cell.Biosens Bioelectron.2011;26(7):3233-93239.
[36] Bunimovich YL,Shin YS,Yeo WS,et al.Quantitative real-time measurements of DNA hybridization with alkylated nonoxidized silicon nanowires in electrolyte solution.J Am Chem Soc.2006;128(50):16323-16331.
[37] Park I,Li Z,Pisano AP,et al.Selective surface functionalization of silicon nanowires via nanoscale joule heating.Nano Lett. 2007;7(10):3106-3111.
[38] Patolsky F,Zheng G,Lieber CM.Fabrication of silicon nanowire devices for ultrasensitive, label-free, real-time detection of biological and chemical species.Nat Protocols.2006;1: 1711-1724.
[39] Stern E.Label-free sensing with semiconducting nanowires. Yale University, 2007.
[40] Stern E,Wagner R,Sigworth FJ,et al.Importance of the Debye screening length on nanowire field effect transistor sensors. Nano Lett.2007;7:3405-3409.
[41] Zhang GJ,Zhang G,Chua JH,et al.DNA sensing by silicon nanowire: charge layer distance dependence.Nano Lett. 2008;8(4):1066-1070.
[42] Sorgenfrei S,Chiu C,Johnston M,et al.Debye screening in single-molecule carbon nanotube field-effect sensors.Nano Lett.2011;11(9):3739-3743.
[43] Zheng G,Gao XPA,Lieber CM.Frequency domain detection of biomolecules using silicon nanowire biosensors.Nano Lett. 2010;10:3179-3183.
[44] Pui TS,Agarwal A,Ye F,et al.Nanoelectronic detection of triggered secretion of pro-inflammatory cytokines using CMOS compatible silicon nanowires.Biosens Bioelectron. 2011;26(5):2746-2750.
[45] Lee MH,Lee K,Jung SW. Multiplexed detection of protein markers with silicon nanowire FET and sol-gel matrix[C]//Engineering in Medicine and Biology Society (EMBC), 2012 Annual International Conference of the IEEE. IEEE, 2012: 570-573.
[46] Ivanov YD,Pleshakova TO,Kozlov AF.SOI nanowire for the high-sensitive detection of HBsAg and a-fetoprotein.Lab Chip.2012;12:5104-5111.
[47] Vickers AJ,Savage C,O’Brien MF,et al.Systematic review of pretreatment prostate-specific antigen velocity and doubling time as predictors for prostate cancer.J Clin Oncol.2009; 27:398-403.
[48] Shariat SF,Scardino PT,Lilja H.Screening for prostate cancer: an update.Can J Urol.2008;15(6):4363-4374.
[49] Rubach M,Szymendera JJ,Kaminska J,et al. Serum CA 15.3, CEA and ESR patterns in breast cancer.Int J Biol Markers. 1997;12(4):168-173.
[50] Uehara M,Kinoshita T,Hojo T.Long-term prognostic study of carcinoembryonic antigen(CEA) and carbohydrate antigen 15-3 (CA 15-3) in breast cancer.Int J ClinOncol.2008;13: 447-451.
[51] Saito S,Ojima H,Ichikawa H,et al.Molecular background of α-fetoprotein in liver cancer cells as revealed by global RNA expression analysis.Cancer Sci.2008;99:2402-2409.
[52] Dvorak HF,Detmar M,Claffey KP,et al.Vascular permeability factor/vascular endothelial growth factor: an important mediator of angiogenesis in malignancy and inflammation.Int Arch Allergy Immunol.1995;107(1-3): 233-235.
[53] Mori A,Arii S,Furutani M,et al.Soluble Flt-1 gene therapy for peritoneal metastases using HVJ-cationic liposomes.Gene Therapy.2000;7(12): 1027-1033. |
| [1] | He Yan-ping, Ma De-chun, Li Lei, Zhang Li, Zheng Shuang, Dong Ke-xin. Hemocompatibility and surface modification of artificial blood vessel materials [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1272-1276. |
| [2] | Wu Qiong, Hu Wei-ping. Clinical common problems after restoration with telescopic crown-retained removable partial dentures [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1301-1305. |
| [3] | Feng Chao, Li Zhe, Lv Xiang-guo, Xu Yue-min, Fu Qiang. In vitro preparation and biochemical evaluation of oxygen generative keratin/silk fibroin compound biomaterial [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8480-8486. |
| [4] | Li Tian-shi, Zeng Wen-ni, He Jun-jun. Chitin and its derivatives inhibit scar formation [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8504-8508. |
| [5] | Li Bing-ting, Jia Ying-zhen, Liu Zhi-fang, Song Yuan, Hou Xiao-wei. Effect of freeze-dried bone xenograft and platelet-rich fibrin compound on osteogenesis and osseointegration of alveolar bone defects [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8376-8381. |
| [6] | Wang Xiao-dong, Zhang Yong-hong. Preparation and performance of recombinant human bone morphogenetic protein-2-poly(hydroxybutyrate-co-hydroxyoctanoate) nanospheres [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8405-8408. |
| [7] |
Wang Xue-ming, Xing Chang-min, Li Nan.
Preparation methods and in vitro release of Resina Draconis-loaded biodegradable polymeric hollow nanoparticles
|
| [8] | Ma Song-feng, Cao Hui, Zheng Feng, Qiao Jun, Zhang Guo-ming. Concomitant cardiac valve replacement and coronary artery bypass grafting [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(5): 699-704. |
| [9] | Ruan Guang-ping, Yao Xiang, Liu Ju-fen, Shu Fan, He Jie, Yang Jian-yong, Pang Rong-qing, Pan Xing-hua. Role of human umbilical cord mesenchymal stem cells: cell transplantation, immuoregulation and target cells [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(41): 6714-6718. |
| [10] | Lu Hua-ding, Lian Li-yi, Chen Ming-wei, Dai Yu-hu. Cloning and expression of Asperguillus endo-chitosanase gene in Escherichia coli [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(34): 5490-5496. |
| [11] | Fan Zhong-bao, Rong Da-qing, Liu Qing-feng. Prolene hernia system versus polypropylene mesh plug in tension-free hernia repair [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(34): 5535-5539. |
| [12] | Li A-li. Absorbable ligating clip in laparoscopic hysterectomy [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(34): 5561-5565. |
| [13] | Zhao Hua-xiang, Zhao Shan-mei, Xin Xin, Zhang Bo, Ma Ning-hu, Li Mu-jia, Zhang Meng-qi, Li Ang . Inflamed dental pulp stem cells: initial research and future development [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(23): 3756-3761. |
| [14] | Wang Jin-ning, Pi Bin, Wang Peng, Zhu Xue-song, Yang Hui-lin. Cytocompatibility and cytotoxicity of a novel composite bone cement material composed of chitosan microsphere, alpha-tricalcium phosphate and silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(16): 2519-2525. |
| [15] | Chen Peng, Zhang Jie, Rong Dong-ming, Han Zhong-yu, Yuan Si-jie, Tian Jing. Effects of non-dextran coated superparamagnetic iron oxide nanoparticles on proliferation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(16): 2526-2531. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||



