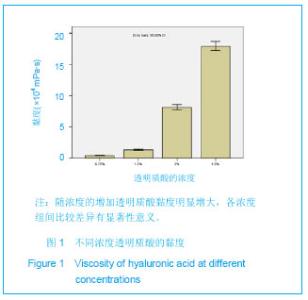
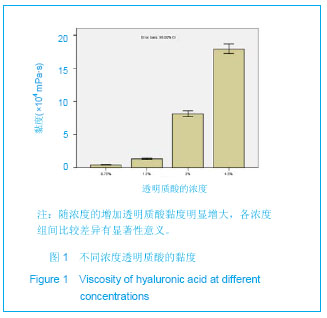
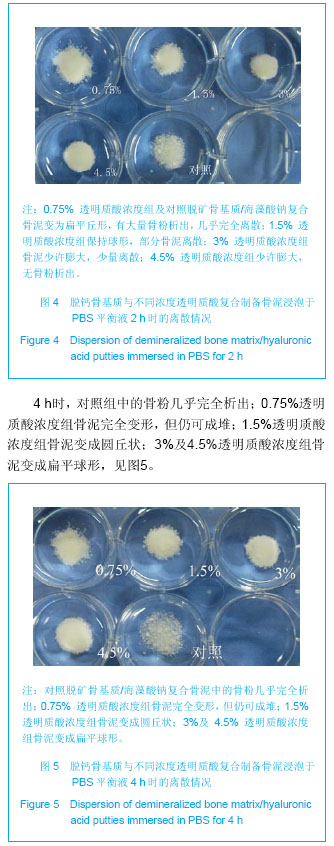
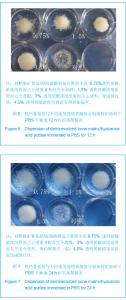
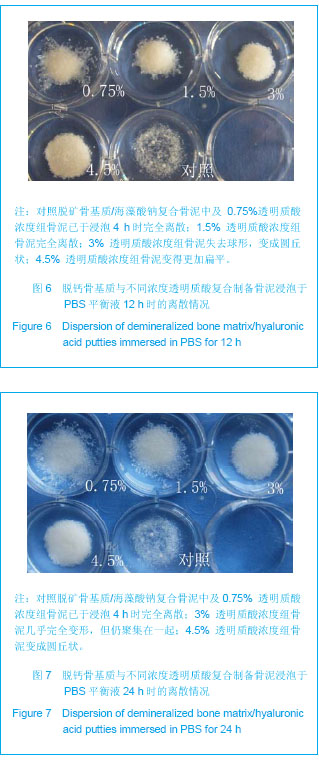
Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (47): 8182-8188.doi: 10.3969/j.issn.2095-4344.2013.47.007
Previous Articles Next Articles
Preparation and cell compatibility of malleable demineralized bone matrix/hyaluronic acid putty
Zhang Nai-li, Zhang Yu-min, Zhou Mo, Yang Ting, Wang Xu-sheng, Ma Shao-ying, Xu Wei-jun, Dong Li,Li Bao-xing
- Medical Tissue Bank, China Institute for Radiation Protection, Taiyuan 030006, Shanxi Province, China
-
Revised:2013-08-18Online:2013-11-19Published:2013-11-19 -
Contact:Li Bao-xing, Investigator, Doctoral supervisor, Medical Tissue Bank, China Institute for Radiation Protection, Taiyuan 030006, Shanxi Province, China zymsir@sina.com -
About author:Zhang Nai-li☆, Studying for doctorate, Assistant investigator, Medical Tissue Bank, China Institute for Radiation Protection, Taiyuan 030006, Shanxi Province, China znl-0@163.com -
Supported by:the Youth Science and Technology Foundation of China Institute for Radiation Protection, No. YQ120807*
CLC Number:
Cite this article
Zhang Nai-li, Zhang Yu-min, Zhou Mo, Yang Ting, Wang Xu-sheng, Ma Shao-ying, Xu Wei-jun, Dong Li,Li Bao-xing. Preparation and cell compatibility of malleable demineralized bone matrix/hyaluronic acid putty[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(47): 8182-8188.
share this article
| [1] Liao SS,Guan K,Cui FZ,et al.Lumbar spinal fusion with a mineralized collagen matrix and rhBMP-2 in a rabbit model.Spine (Phila Pa 1976). 2003;28(17):1954-1960. [2] Baroth S,Bourges X,Goyenvalle E,et al.Injectable biphasic calcium phosphate bioceramic: The HYDROS concept. Biomed Mater Eng.2009; 19(1):71-76. [3] Wang Z,Lu B,Chen L,et al. Evaluation of an osteostimulative putty in the sheep spine.J Mater Sci Mater Med.2011; 22(1): 185-191. [4] Bodakhe S,Verma S,Garkhal K,et al.Injectable photocrosslinkable nanocomposite based on poly(glycerol sebacate) fumarate and hydroxyapatite: development, biocompatibility and bone regeneration in a rat calvarial bone defect model. Nanomedicine (Lond).2013. [Epub ahead of print] [5] 张乃丽,李宝兴,张育敏,等.可塑形骨泥的研究进展[J].中国修复重建外科杂志, 2012,26(11):1391-1397. [6] Becker W, Burton E, Raul C. A comparison of demineralized freeze-dried bone and autologous bone to induce bone formation in human extraction sockets. J Periodontol. 1994; 65(12):1128-1133. [7] 孙晓雷,马信龙,马剑雄,等. 牛脱细胞骨基质的生物力学特性及其黏附性[J].中国组织工程研究与临床康复,2008,12(49): 9659-9663. [8] Urist MR,Chang JJ,Lietze A,et al.Preparation and bioassay of bone morphogenetic protein and polypeptide fragments. Methods Enzymol.1987; 146:294-312. [9] Lee DW, Koo KT, Seol YJ, et al. Bone regeneration effects of human allogenous bone substitutes: A preliminary study. J Periodontal Implant Sci. 2010; 40:132-138. [10] Aspriello SD,Ferrante L,Rubini C,et al.Comparative study of DFDBA in combination with enamel matrix derivative versus DFDBA alone for treatment of periodontal intrabony defects at 12 months post-surgery.Clin Oral Investig.2011; 15:25-32. [11] Pietrzak WS,Ali SN,Chitturi D, et al. BMP depletion occurs during prolonged acid demineralization of bone: Characterization and implications for graft preparation.Cell Tissue Bank.2011;12:81-88. [12] Darby I. Periodontal materials.Aust Dent J.2011; 56:107-118. [13] Chung C,Beecham M,Mauck RL,et al.The influence of degradation characteristics of hyaluronic acid hydrogels on in vitro neocartilage formation by mesenchymal stem cells. Biomaterials.2009;30(26): 4287-4296. [14] Ibrahim S, Kang QK, Ramamurthi A. The impact of hyaluronic acid oligomer content on physical, mechanical, and biologic properties of divinyl sulfone-crosslinked hyaluronic acid hydrogels. J Biomed Mater Res A. 2010; 94(2): 355-370. [15] Dereure O. Hyaluronic acid and immunity. Ann Dermatol Venereol. 2010; 137 Suppl 1:S26-29. [16] Mendoza G,Alvarez AI,Pulido MM,et al.Inhibitory effects of different antioxidants on hyaluronan depolymerization. Carbohydr Res.2007;342(1):96-102. [17] Jae KK,Periasamy S,Jae HK,et al. Structural and antioxidant properties of gamma irradiated hyaluronic acid. Food Chem. 2008;109(4):763-770. [18] Day AJ,de la Motte CA.Hyaluronan cross-linking: a protective mechanism in inflammation? Trends Immunol.2005;26(12): 637-643. [19] Termeer C,Benedix F,Sleeman J,et al.Oligosaccharides of Hyaluronan activate dendritic cells via toll-like receptor 4.J Exp Med.2002;195(1):99-111. [20] Kobayashi H,Sun GW,Terao T.Production of prostanoids via increased cyclo-oxygenase-2 expression in human amnion cells in response to low molecular weight hyaluronic acid fragment.Biochim Biophys Acta. 1998;1425(2):369-376. [21] Jiang D,Liang J,Noble PW.Regulation of non-infectious lung injury, inflammation, and repair by the extracellular matrix glycosaminoglycan hyaluronan. Anat Rec (Hoboken).2010; 293(6):982-985. [22] Franz S, Allenstein F, Kajahn J, et al. Artificial extracellular matrices composed of collagen I and high-sulfated hyaluronan promote phenotypic and functional modulation of human pro-inflammatory M1 macrophages. Acta Biomater. 2013; 9(3):5621-5629. [23] West DC, Hampson IN, Arnold F, et al. Angiogenesis induced by degradation products of hyaluronic acid. Science.1985; 228(4705):1324-1326. [24] Skardal A, Zhang J, McCoard L, et al. Photocrosslinkable hyaluronan-gelatin hydrogels for two-step bioprinting. Tissue Eng Part A.2010; 16(8):2675-2685. [25] Urist MR. Bone:formation by autoinduction. Science. 1965; 150:893-899. [26] GB/T 16886.12-2005. 医疗器械生物学评价,第12部分:样品制备与参照样品. [27] GB/T 16886.5-2003. 医疗器械生物学评价,第5部分:体外细胞毒性试验 [28] Kneserl U,Schaeferl DJ,Munder B,et al.Tissue engineering of bone. Min Invas Ther&Allied Technol. 2002;11(3):107-116. [29] Innes JF,Myint P. Dminerialised bone matrix in veterinary orthopaedics: a review. Vet Comp Orthop Traumatol.2010; 23(6):393-399. [30] Lee YP, Jo M, Luna M, et al.The efficacy of different commercially available demineralized bone matrix substances in an athymic rat model. J Spinal Disord Tech. 2005;18(5):439-444. [31] 徐伟俊,李宝兴,张育敏,等.可塑型脱矿骨基质/海藻酸钠复合材料的制备及细胞相容性[J].中国组织工程研究,2012,16(29): 5433-5436. [32] Wang JC,Kanim LE,Nagakawa IS,et al.Dose-dependent toxicity of a commercially available demineralized bone matrix material. Spine (Phila Pa 1976).2001;26(13):1429-1436. [33] Prestwich GD. Hyaluronic acid-based clinical biomaterials derived for cell and molecule delivery in regenerative medicine.J Control Relaese.2011;155(2):193-199. [34] Wallin RF, Arscott EF.A Practical Guide to ISO 10993-5: Cytotoxicity. MDDI. 1998; 20:96-98. [35] Galia CR,Macedo CA,Rosito R,et al.In vitro and in vivo evaluation of lyophilized bovine bone biocompatibility.Clinics (Sao Paulo). 2008;63(3):801-806. [36] Ishiyama M,Tominaga H,Shiga M,et al.A combined assay of cell viability and in vitro cytotoxicity with a highly water-soluble tetrazolium salt, neutral red and crystal violet.Biol Pharm Bull. 1996;19(11):1518-1520. [37] Miyamoto T, Min W, Lillehoj HS. Lymphocyte proliferation response during Eimeria tenella infection assessed by a new, reliable, nonradioactive colorimetric assay. Avian Dis. 2002; 46(1):10-16. |
| [1] | He Yan-ping, Ma De-chun, Li Lei, Zhang Li, Zheng Shuang, Dong Ke-xin. Hemocompatibility and surface modification of artificial blood vessel materials [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(8): 1272-1276. |
| [2] | Li Bing-ting, Jia Ying-zhen, Liu Zhi-fang, Song Yuan, Hou Xiao-wei. Effect of freeze-dried bone xenograft and platelet-rich fibrin compound on osteogenesis and osseointegration of alveolar bone defects [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8376-8381. |
| [3] | Wang Xiao-dong, Zhang Yong-hong. Preparation and performance of recombinant human bone morphogenetic protein-2-poly(hydroxybutyrate-co-hydroxyoctanoate) nanospheres [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8405-8408. |
| [4] | Feng Chao, Li Zhe, Lv Xiang-guo, Xu Yue-min, Fu Qiang. In vitro preparation and biochemical evaluation of oxygen generative keratin/silk fibroin compound biomaterial [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8480-8486. |
| [5] | Li Tian-shi, Zeng Wen-ni, He Jun-jun. Chitin and its derivatives inhibit scar formation [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8504-8508. |
| [6] | Yan Hao-ran, Cai Jun-feng, Shen Bin, Wen Jun-xiang, Tan Jun. Sodium hyaluronate for treatment of chondromalacia patella syndrome [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(52): 8525-8528. |
| [7] | Ma Song-feng, Cao Hui, Zheng Feng, Qiao Jun, Zhang Guo-ming. Concomitant cardiac valve replacement and coronary artery bypass grafting [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(5): 699-704. |
| [8] | Lu Hua-ding, Lian Li-yi, Chen Ming-wei, Dai Yu-hu. Cloning and expression of Asperguillus endo-chitosanase gene in Escherichia coli [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(34): 5490-5496. |
| [9] | Fan Zhong-bao, Rong Da-qing, Liu Qing-feng. Prolene hernia system versus polypropylene mesh plug in tension-free hernia repair [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(34): 5535-5539. |
| [10] | Li A-li. Absorbable ligating clip in laparoscopic hysterectomy [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(34): 5561-5565. |
| [11] | Huang Xiao-nan. Percutaneous vertebroplasty for treatment of osteoporotic vertebral fractures: high-viscosity versus low-viscosity bone cement [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(16): 2461-2467. |
| [12] | Liu Li-xia, Chen Lin. Comparison of fluoride release and solubility for different glass ionomer cements [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(16): 2480-2486. |
| [13] | Zhou Zheng, Yang Ze-hui, He Hui-yu, Cui Jie. Biocompatibility of freeze-dried antigen-extracted sheep cancellous bone scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(16): 2499-2505. |
| [14] | Wang Jin-ning, Pi Bin, Wang Peng, Zhu Xue-song, Yang Hui-lin. Cytocompatibility and cytotoxicity of a novel composite bone cement material composed of chitosan microsphere, alpha-tricalcium phosphate and silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(16): 2519-2525. |
| [15] | Chen Peng, Zhang Jie, Rong Dong-ming, Han Zhong-yu, Yuan Si-jie, Tian Jing. Effects of non-dextran coated superparamagnetic iron oxide nanoparticles on proliferation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(16): 2526-2531. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||