Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (17): 2761-2766.doi: 10.3969/j.issn.2095-4344.3197
Previous Articles Next Articles
Exercise improves progression of Alzheimer’s disease in mice: a dose-effect relationship
Zhang Xiang, Zhang Yeting
- School of Physical Education, Chengdu University, Chengdu 610106, Sichuan Province, China
-
Received:2020-04-27Revised:2020-04-28Accepted:2020-05-22Online:2021-06-18Published:2021-01-08 -
Contact:Zhang Yeting, PhD, Assistant, School of Physical Education, Chengdu University, Chengdu 610106, Sichuan Province, China -
About author:Zhang Xiang, Master, Associate professor, School of Physical Education, Chengdu University, Chengdu 610106, Sichuan Province, China -
Supported by:Sichuan Provincial Science and Technology Project, No. 2020YFH0184 (to ZYT)
CLC Number:
Cite this article
Zhang Xiang, Zhang Yeting. Exercise improves progression of Alzheimer’s disease in mice: a dose-effect relationship[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(17): 2761-2766.
share this article
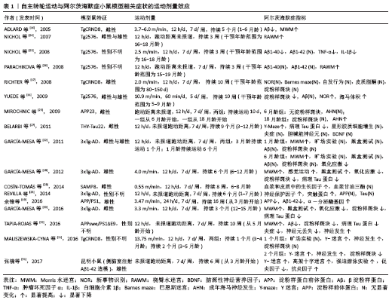
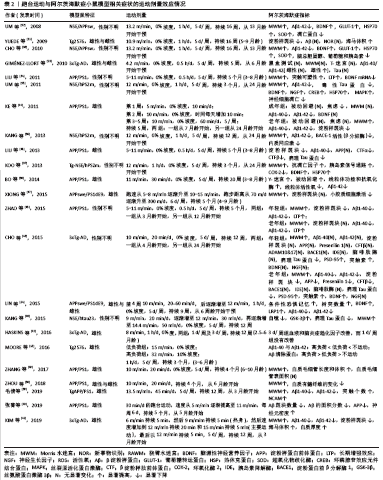
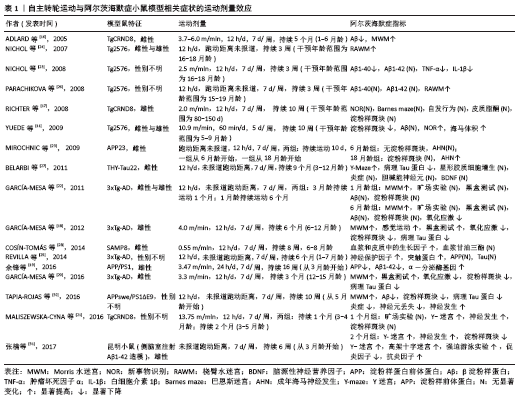
2.1 运动模式对阿尔茨海默症小鼠模型的影响 运动对阿尔茨海默症动物模型影响的研究主要采用2种运动模式:自主转轮运动(自主运动)与跑台运动(强迫运动)。跑台运动被认为是典型的强迫运动,而这种运动形式被证明会增加啮齿类动物应激反应的生物标志物[9]。在跑台运动中,应激压力可能是由于跑台后面的电击刺激引起的,或者是由于必须以预定的速度跑步引起的,也可能两者兼有。有学者研究发现,应激压力可能会加重小鼠模型的阿尔茨海默症病理过程,因此确定应激压力对强迫运动研究的影响非常重要[10]。通过跑台运动进行干预能够较为精确地控制运动强度和持续时间,这是自主转轮运动干预所不能实现的;然而,跑台运动所产生的应激压力可能会抵消一些运动所带来的益处。作者推测,与跑台运动相比,自主转轮运动属于自主运动,可能会对模型鼠产生更小的应激压力,而通过跑台运动和自主转轮运动的比较则能够较为深入了解不同运动模式及运动量对阿尔茨海默症生物标志物的剂量关系。有学者通过对阿尔茨海默症模型小鼠分别进行为期16周的跑台运动与自主转轮运动干预发现,运动小鼠的海马体积都大于不运动小鼠,而自主转轮运动干预小鼠具有更好的行为表现及更少的淀粉样蛋白沉积[11]。跑台运动组的小鼠没有获得与自主转轮运动组小鼠相同的益处,这表明运动模式之间存在一定的差异,这可能是跑台运动产生的应激压力导致了差异的存在,特别是在认知方面,自主转轮运动似乎更有优势。另外,在搜集的研究中,也有很少部分研究对阿尔茨海默症小鼠采用游泳的运动方式进行干预,但由于游泳的运动强度较难把握,且很难与跑步的运动强度做对比,因此这部分研究未纳入该综述中。 2.2 运动改善阿尔茨海默症病程的剂量效应 运动干预阿尔茨海默症模型鼠的研究结果大多为阳性,但是也有研究报道称,运动干预对阿尔茨海默症模型鼠的病理特征或认知改善等方面几乎没有影响[6,12-13]。在这些研究中,很难判断阿尔茨海默症模型鼠接受了多少运动刺激。迄今为止,只有很少的研究评估了运动对阿尔茨海默症模型鼠的剂量效应[14-15]。可以想象,在动物模型中,如果运动强度、持续时间和频率低于一个临界阈值,那么跑台运动所带来的应激压力很可能会抵消运动所带来的有益影响。表1中展示的是自主转轮运动与阿尔茨海默症小鼠模型相关症状的运动剂量效应情况[11,16-31],表2中展示的是跑台运动与阿尔茨海默症小鼠模型相关症状的运动剂量效应情况[11,13-15,32-50]。 2.2.1 自主转轮运动与阿尔茨海默病症状的剂量效应 自主转轮运动是由动物本身自发运动来推动跑轮转动,在这种运动中,笼内动物长期活动的信息能够被记录下来,如跑轮转动方向、转数、累计总行程等,但是很难准确地确定运动强度。因此作者试图根据表1所示的阿尔茨海默症动物模型中自主转轮运动研究的参数来估计运动强度。根据研究中所报道的跑动距离及跑动时间,可以估算出动物跑动的速度(跑动速度=跑动距离/跑动时间)。根据估算,所纳入的研究中模型鼠运动的速度范围较大,在0.55-13.75 m/min之间,然而这很可能低估了模型鼠的速度。比如,如果让模型鼠在含有自主转轮的笼子中自由活动12 h,而模型鼠只在自主转轮上运动了4 h,那么模型鼠的跑动速度将高于估算结果。有研究发现,在对Tg2576小鼠进行为期4个月,每天1 h的自主转轮运动干预过程中,小鼠的跑动速度平均为10.9 m/min[11]。综合而言,运动速度超过2.5 m/min的研究显示其动物模型的阿尔茨海默症病理学指标如β淀粉样蛋白或Tau蛋白得到了改善,认知行为学测试有所好转[11,16-21],并且其改善效果随运动速度的增加而增加。当然,由于可能存在对运动速度的低估,模型鼠实际的自主转轮运动速度会大于2.5 m/min,为了能够更好地确定其运动速度,这就需要在研究过程中更加详细记录模型鼠各时段的数据。除了跑动速度,持续时间也是影响运动剂量的关键,有研究分别对TgCRND8阿尔茨海默症小鼠进行为期1个月和为期2个月的自主转轮运动干预,发现持续2个月的运动干预能够更好地改善阿尔茨海默症小鼠病程[21]。但也有研究发现,通过对3xTg-AD小鼠进行为期1个月和为期6个月的自主转轮运动干预,发现两组的阿尔茨海默症病程改善并没有显著差别,都只是在认知行为学方面起到了改善的作用,对于阿尔茨海默症小鼠的病理也没有改善作用[22]。由于该研究未报道小鼠跑动的距离,无法估算小鼠的跑动速度,因此作者推测该研究中小鼠的跑动速度可能比较低,因此造成了运动效果的不佳。另外,起始干预的年龄也可能成为运动起到改善阿尔茨海默症病程的关键,有学者对6月龄和18月龄的APP23阿尔茨海默症小鼠进行自主转轮运动干预,发现18月龄小鼠的成年海马神经发生显著增加,而6月龄小鼠并未出现,这可能是由于18月龄APP23阿尔茨海默症小鼠具有了显著的阿尔茨海默症病理特征,此时进行运动干预可能效果更好[23]。 2.2.2 跑台运动与阿尔茨海默病症状的剂量效应 运动强度在跑台运动中可以通过调整跑速、跑动时间、频率等进行控制,因此可以直接比较表2所列研究中不同运动强度对阿尔茨海默症的影响。凡是运动速度超过每天10 m/min的研究,都报道了运动对阿尔茨海默症小鼠模型的病理或认知影响的改善作用[11,13-15,32-41]。如果跑动速度低于10 m/min,则很难对阿尔茨海默症小鼠起到改善作用。比如一项对3xTg-AD小鼠进行跑速为4.2 m/min的干预研究,发现运动虽然对雄性小鼠的Aβ40/42比率有一定的改善作用,但是对其他指标均没有显著改善作用,表明运动强度可能在改善阿尔茨海默症病理过程中具有重要的影响力[13]。那么在达到了10 m/min的运动速度后,是否会随着运动强度的增加,阿尔茨海默症小鼠的病理改善也会加强呢?一项对Tg2576 AD小鼠的研究发现,采用较高负荷(32 m/min,10%坡度)进行干预的阿尔茨海默症小鼠的病理及认知改善都优于采用较低负荷(15 m/min,0%坡度)进行干预的阿尔茨海默症小鼠[15]。分别以10,15, 18 m/min对3月龄APP/PS1小鼠进行5 d/周,长达5个月的跑台运动干预后发现,10 m/min的跑速虽然可以降低阿尔茨海默症小鼠的Aβ1-42沉积,但并不足以改善阿尔茨海默症小鼠的病程;而15 m/min和18 m/min的跑速均可以显著降低阿尔茨海默症小鼠Aβ1-42的沉积,增加其成年海马神经发生,改善其学习记忆能力;其中15 m/min的跑速起到的改善效果最佳(该研究还未发表)。因此,作者推测很可能在一定的运动负荷范围内,阿尔茨海默症小鼠的病理改善会随运动强度的增加而增大。除了跑动速度,运动的频率、时间似乎也是影响运动剂量的关键,有研究对3xTG-AD小鼠进行较低强度 (8 m/min)的运动干预研究发现,在12周内每周运动3次则可以改善相关的炎症指标,而每周只运动1次则没有改善效果,这表明运动频率起到了重要的作用[42]。另外,起始干预的年龄也可能成为运动起到改善阿尔茨海默症病程的关键,一项对4月龄和24月龄的3xTg-AD小鼠进行跑台运动干预研究发现,运动对24月龄AD小鼠的病理改善作用显著优于4月龄AD小鼠[43]。但是也有研究发现,起始干预的年龄并不是运动起到改善阿尔茨海默症病程的关键,如对7月龄和24月龄的APP/PS1阿尔茨海默症小鼠进行跑台运动干预研究发现,运动对这两种年龄的阿尔茨海默症小鼠都有很好的改善作用[44]。另一项研究通过对3月龄和12月龄的APP/PS1阿尔茨海默症小鼠进行跑台运动干预研究也发现,运动对这两种年龄的阿尔茨海默症小鼠都有很好的改善作用[45]。这种不一致的研究结果很可能与阿尔茨海默症小鼠的种系及转基因造模方式不同有关。 表1和表2总结了以往关于运动对阿尔茨海默症小鼠病理和认知影响的相关研究,这些研究较为直观地展现了运动强度对阿尔茨海默症小鼠的影响,尤其是跑台运动(强迫运动)的影响。当然,由于这些研究所采用的阿尔茨海默症小鼠模型不尽相同,而不同模型小鼠的运动能力不一样,因此在比较这些研究的运动强度时就变得比较困难。比如,有研究发现APP/PS1 阿尔茨海默症小鼠中大多数能够以24 m/min的速度、10%的坡度跑1 h,而Tg2576 阿尔茨海默症小鼠在相同的情况下能够以32 m/min的速度奔跑[11]。虽然运动强度确实对阿尔茨海默症小鼠病理产生了有益影响,但仍存在一个问题,即运动强度的阈值是多少?通过对以上研究,尤其是针对跑台运动干预的研究,作者认为运动速度的阈值大约可能是10 m/min。而运动持续时间和频率似乎越大越好,在所列研究中自主转轮运动干预不低于12 h/d,7 d/周,持续3周;跑台运动干预不低于5 d/周,持续9周。 "
| [1] MARTIN P, ANDERS W. World Alzheimer Report 2016. Alzheimer’s Disease International,2016. [2] BROOKMEYER R, JOHNSON E, ZIEGLER-GRAHAM K, et al. Forecasting the global burden of Alzheimer’s disease. Alzheimers Dement. 2007;3(3):186-191. [3] PRINCE M, BRYCE R, ALBANESE E, et al. The global prevalence of dementia: a systematic review and metaanalysis. Alzheimers Dement. 2013;9(1):63-75.e2. [4] KENNEDY G, HARDMAN RJ, MACPHERSON H, et al. How Does Exercise Reduce the Rate of Age-Associated Cognitive Decline? A Review of Potential Mechanisms. J Alzheimers Dis. 2017;55(1):1-18. [5] LIANG KY, MINTUN MA, FAGAN AM, et al. Exercise and Alzheimer’s disease biomarkers in cognitively normal older adults. Ann Neurol. 2010;68(3):311-318. [6] WOLF SA, KRONENBERG G, LEHMANN K, et al. Cognitive and physical activity differently modulate disease progression in the amyloid precursor protein (APP)-23 model of Alzheimer’s disease. Biol Psychiatry. 2006;60(12):1314-1323. [7] 朱为模.运动处方的过去、现在与未来[J].体育科研,2020,41(1):1-18. [8] EIJSVOGELS TM, MOLOSSI S, LEE DC, et al. Exercise at the Extremes: The Amount of Exercise to Reduce Cardiovascular Events. J Am Coll Cardiol. 2016;67(3):316-329. [9] SVENSSON M, ROSVALL P, BOZA-SERRANO A, et al. Forced treadmill exercise can induce stress and increase neuronal damage in a mouse model of global cerebral ischemia. Neurobiol Stress. 2016;5:8-18. [10] BAGLIETTO-VARGAS D, CHEN Y, SUH D, et al. Short-term modern life-like stress exacerbates Aβ-pathology and synapse loss in 3xTg-AD mice. J Neurochem. 2015;134(5):915-926. [11] YUEDE CM, ZIMMERMAN SD, DONG H, et al. Effects of voluntary and forced exercise on plaque deposition, hippocampal volume, and behavior in the Tg2576 mouse model of Alzheimer’s disease. Neurobiol Dis. 2009;35(3):426-432. [12] CRACCHIOLO JR, MORI T, NAZIAN SJ, et al. Enhanced cognitive activity--over and above social or physical activity--is required to protect Alzheimer’s mice against cognitive impairment, reduce Abeta deposition, and increase synaptic immunoreactivity. Neurobiol Learn Mem. 2007;88(3):277-294. [13] GIMÉNEZ-LLORT L, GARCÍA Y, BUCCIERI K, et al. Gender-Specific Neuroimmunoendocrine Response to Treadmill Exercise in 3xTg-AD Mice. Int J Alzheimers Dis. 2010;2010:128354. [14] LIN TW, SHIH YH, CHEN SJ, et al. Running exercise delays neurodegeneration in amygdala and hippocampus of Alzheimer’s disease (APP/PS1) transgenic mice. Neurobiol Learn Mem. 2015;118:189-197. [15] MOORE KM, GIRENS RE, LARSON SK, et al. A spectrum of exercise training reduces soluble Aβ in a dose-dependent manner in a mouse model of Alzheimer’s disease. Neurobiol Dis. 2016;85:218-224. [16] ADLARD PA, PERREAU VM, POP V, et al. Voluntary exercise decreases amyloid load in a transgenic model of Alzheimer’s disease. J Neurosci. 2005;25(17):4217-4221. [17] RICHTER H, AMBRÉE O, LEWEJOHANN L, et al. Wheel-running in a transgenic mouse model of Alzheimer’s disease: protection or symptom? Behav Brain Res. 2008;190(1):74-84. [18] GARCÍA-MESA Y, GIMÉNEZ-LLORT L, LÓPEZ LC, et al. Melatonin plus physical exercise are highly neuroprotective in the 3xTg-AD mouse. Neurobiol Aging. 2012;33(6):1124.e13-29. [19] 余锋,徐波,季浏.α-分泌酶在自主运动调节APP/PS1转基因小鼠海马APP与Aβ42中的作用研究[J].体育科学,2016,36(7):49-55. [20] GARCÍA-MESA Y, COLIE S, CORPAS R, et al. Oxidative Stress Is a Central Target for Physical Exercise Neuroprotection Against Pathological Brain Aging. J Gerontol A Biol Sci Med Sci. 2016;71(1):40-49. [21] MALISZEWSKA-CYNA E, XHIMA K, AUBERT I. A Comparative Study Evaluating the Impact of Physical Exercise on Disease Progression in a Mouse Model of Alzheimer’s Disease. J Alzheimers Dis. 2016;53(1):243-257. [22] GARCÍA-MESA Y, LÓPEZ-RAMOS JC, GIMÉNEZ-LLORT L, et al. Physical exercise protects against Alzheimer’s disease in 3xTg-AD mice. J Alzheimers Dis. 2011; 24(3):421-454. [23] MIROCHNIC S, WOLF S, STAUFENBIEL M, et al. Age effects on the regulation of adult hippocampal neurogenesis by physical activity and environmental enrichment in the APP23 mouse model of Alzheimer disease. Hippocampus. 2009;19(10):1008-1018. [24] NICHOL KE, PARACHIKOVA AI, COTMAN CW. Three weeks of running wheel exposure improves cognitive performance in the aged Tg2576 mouse. Behav Brain Res. 2007;184(2):124-132. [25] NICHOL KE, POON WW, PARACHIKOVA AI, et al. Exercise alters the immune profile in Tg2576 Alzheimer mice toward a response coincident with improved cognitive performance and decreased amyloid. J Neuroinflammation. 2008;5:13. [26] PARACHIKOVA A, NICHOL KE, COTMAN CW. Short-term exercise in aged Tg2576 mice alters neuroinflammation and improves cognition. Neurobiol Dis. 2008;30(1):121-129. [27] BELARBI K, BURNOUF S, FERNANDEZ-GOMEZ FJ, et al. Beneficial effects of exercise in a transgenic mouse model of Alzheimer’s disease-like Tau pathology. Neurobiol Dis. 2011;43(2):486-494. [28] COSÍN-TOMÁS M, ALVAREZ-LÓPEZ MJ, SANCHEZ-ROIGE S, et al. Epigenetic alterations in hippocampus of SAMP8 senescent mice and modulation by voluntary physical exercise. Front Aging Neurosci. 2014;6:51. [29] REVILLA S, SUÑOL C, GARCÍA-MESA Y, et al. Physical exercise improves synaptic dysfunction and recovers the loss of survival factors in 3xTg-AD mouse brain. Neuropharmacology. 2014;81:55-63. [30] TAPIA-ROJAS C, ARANGUIZ F, VARELA-NALLAR L, et al. Voluntary Running Attenuates Memory Loss, Decreases Neuropathological Changes and Induces Neurogenesis in a Mouse Model of Alzheimer’s Disease. Brain Pathol. 2016; 26(1):62-74. [31] 张楠,王莉智,杨桂姣,等.自主跑轮运动对阿尔兹海默病模型小鼠认知、情绪以及杏仁核炎性因子表达的影响[J].神经解剖学杂志,2017,33(6):741-747. [32] UM HS, KANG EB, LEEM YH, et al. Exercise training acts as a therapeutic strategy for reduction of the pathogenic phenotypes for Alzheimer’s disease in an NSE/APPsw-transgenic model. Int J Mol Med. 2008;22(4):529-539. [33] CHO JY, UM HS, KANG EB, et al. The combination of exercise training and alpha-lipoic acid treatment has therapeutic effects on the pathogenic phenotypes of Alzheimer’s disease in NSE/APPsw-transgenic mice. Int J Mol Med. 2010;25(3): 337-346. [34] LIU HL, ZHAO G, CAI K, et al. Treadmill exercise prevents decline in spatial learning and memory in APP/PS1 transgenic mice through improvement of hippocampal long-term potentiation. Behav Brain Res. 2011;218(2):308-314. [35] UM HS, KANG EB, KOO JH, et al. Treadmill exercise represses neuronal cell death in an aged transgenic mouse model of Alzheimer’s disease. Neurosci Res. 2011;69(2):161-173. [36] KANG EB, KWON IS, KOO JH, et al. Treadmill exercise represses neuronal cell death and inflammation during Aβ-induced ER stress by regulating unfolded protein response in aged presenilin 2 mutant mice. Apoptosis. 2013;18(11):1332-1347. [37] KOO JH, KWON IS, KANG EB, et al. Neuroprotective effects of treadmill exercise on BDNF and PI3-K/Akt signaling pathway in the cortex of transgenic mice model of Alzheimer’s disease. J Exerc Nutrition Biochem. 2013;17(4):151-160. [38] BO H, KANG W, JIANG N, et al. Exercise-induced neuroprotection of hippocampus in APP/PS1 transgenic mice via upregulation of mitochondrial 8-oxoguanine DNA glycosylase. Oxid Med Cell Longev. 2014;2014:834502. [39] 毛倩. 跑台运动对TgAPP/PS1小鼠海马离子型谷氨酸受体介导的突触可塑性的影响研究[D].上海:华东师范大学,2019. [40] 张菁. 长期运动训练对阿尔茨海默病的神经保护作用和机制研究[M]. 济南:山东大学,2019. [41] KIM D, CHO J, KANG H. Protective effect of exercise training against the progression of Alzheimer’s disease in 3xTg-AD mice. Behav Brain Res. 2019;374: 112105. [42] HASKINS M, JONES TE, LU Q, et al. Early alterations in blood and brain RANTES and MCP-1 expression and the effect of exercise frequency in the 3xTg-AD mouse model of Alzheimer’s disease. Neurosci Lett. 2016;610:165-170. [43] CHO J, SHIN MK, KIM D, et al. Treadmill Running Reverses Cognitive Declines due to Alzheimer Disease. Med Sci Sports Exerc. 2015;47(9):1814-1824. [44] KE HC, HUANG HJ, LIANG KC, et al. Selective improvement of cognitive function in adult and aged APP/PS1 transgenic mice by continuous non-shock treadmill exercise. Brain Res. 2011;1403:1-11. [45] ZHAO G, LIU HL, ZHANG H, et al. Treadmill exercise enhances synaptic plasticity, but does not alter β-amyloid deposition in hippocampi of aged APP/PS1 transgenic mice. Neuroscience. 2015;298:357-366. [46] LIU HL, ZHAO G, ZHANG H, et al. Long-term treadmill exercise inhibits the progression of Alzheimer’s disease-like neuropathology in the hippocampus of APP/PS1 transgenic mice. Behav Brain Res. 2013;256:261-272. [47] XIONG JY, LI SC, SUN YX, et al. Long-term treadmill exercise improves spatial memory of male APPswe/PS1dE9 mice by regulation of BDNF expression and microglia activation. Biol Sport. 2015;32(4):295-300. [48] KANG EB, CHO JY. Effect of treadmill exercise on PI3K/AKT/mTOR, autophagy, and Tau hyperphosphorylation in the cerebral cortex of NSE/htau23 transgenic mice. J Exerc Nutrition Biochem. 2015;19(3):199-209. [49] ZHANG Y, CHAO FL, ZHOU CN, et al. Effects of exercise on capillaries in the white matter of transgenic AD mice. Oncotarget. 2017;8(39):65860-65875. [50] ZHOU CN, CHAO FL, ZHANG Y, et al. Sex Differences in the White Matter and Myelinated Fibers of APP/PS1 Mice and the Effects of Running Exercise on the Sex Differences of AD Mice. Front Aging Neurosci. 2018;10:243. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [3] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [4] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [5] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [6] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [7] | Wang Yongsheng, Wu Yang, Li Yanchun. Effect of acute high-intensity exercise on appetite hormones in adults: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1305-1312. |
| [8] | Wu Xun, Meng Juanhong, Zhang Jianyun, Wang Liang. Concentrated growth factors in the repair of a full-thickness condylar cartilage defect in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1166-1171. |
| [9] | Shen Jinbo, Zhang Lin. Micro-injury of the Achilles tendon caused by acute exhaustive exercise in rats: ultrastructural changes and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1190-1195. |
| [10] | Li Jing, Xie Jianshan, Cui Huilin, Cao Ximei, Yang Yanping, Li Hairong. Expression and localization of diacylglycerol kinase zeta and protein kinase C beta II in mouse back skin with different coat colors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1196-1200. |
| [11] | Chen Jiming, Wu Xiaojing, Liu Tianfeng, Chen Haicong, Huang Chengshuo. Effects of silymarin on liver injury and bone metabolism induced by carbon tetrachloride in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1224-1228. |
| [12] | Tan Jingyu, Liu Haiwen. Genome-wide identification, classification and phylogenetic analysis of Fasciclin gene family for osteoblast specific factor 2 [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1243-1248. |
| [13] | Wang Mengting, Gu Yanping, Ren Wenbo, Qin Qian, Bai Bingyi, Liao Yuanpeng. Research hotspots of blood flow restriction training for dyskinesia based on visualization analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1264-1269. |
| [14] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [15] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||