Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (18): 2921-2926.doi: 10.3969/j.issn.2095-4344.1735
Previous Articles Next Articles
Material composite and properties of magnetic bone cement
- 1Department of Orthopedics, Chinese PLA General Hospital, Beijing 100853, China; 2Department of Orthopedics, Third Affiliated Hospital of Guangxi University of Chinese Medicine, Liuzhou 545001, Guangxi Zhuang Autonomous Region, China
-
Received:2019-03-04Online:2019-06-28Published:2019-06-28 -
Contact:Tang Peifu, Chief physician, Department of Orthopedics, Chinese PLA General Hospital, Beijing 100853, China -
About author:Wang Runsheng, Doctoral candidate, Attending physician, Department of Orthopedics, Chinese PLA General Hospital, Beijing 100853, China; Department of Orthopedics, Third Affiliated Hospital of Guangxi University of Chinese Medicine, Liuzhou 545001, Guangxi Zhuang Autonomous Region, China -
Supported by:the National Natural Science Foundation of China (General Program), No. 51772328 (to MKY); the National Natural Science Foundation of China for the Youth, No. 81702121 (to LJH); the Major Project of Military Logistics, No. AWS17J004 (to LJH); the Clinical Research Support Foundation of Chinese PLA General Hospital, No. 2017FC-TSYS-2006 (to LJH); the First Research Project, No. 2018-4-5014 (to LJH)
CLC Number:
Cite this article
Wang Runsheng, Liu Jianheng, Mao Keya, Tang Peifu. Material composite and properties of magnetic bone cement[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(18): 2921-2926.
share this article
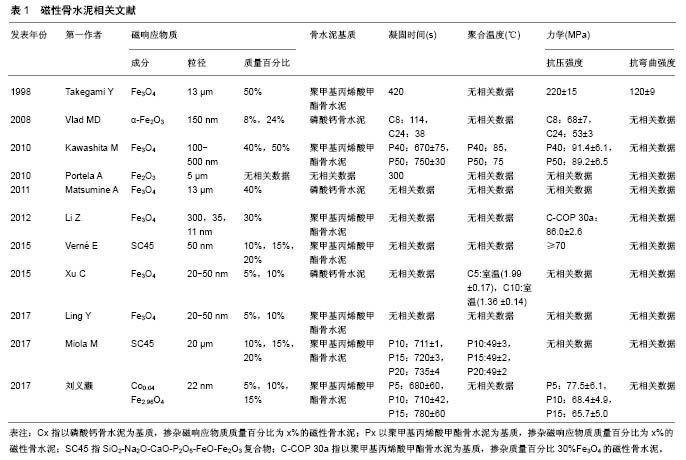
2.1 磁性骨水泥的制备及理化性能 磁性骨水泥由骨水泥基质和磁响应物质2部分组成,骨水泥基质主要为聚甲基丙烯酸甲酯骨水泥[poly(methyl methacrylate),PMMA]和磷酸钙骨水泥。磁响应物质种类的不同,导致磁性骨水泥的力学强度、凝固时间、聚合温度、可注射性等基本参数也不同。Takegami等[8]采用13 µm Fe3O4颗粒与PMMA制成磁性骨水泥,并测试不同质量百分比(10%,20%,40%,50%,80%)磁性骨水泥的力学性能,随着Fe3O4质量百分比的增高,骨水泥的抗压强度和抗弯曲强度逐渐增加。质量百分比50%的磁性骨水泥抗压强度为(220±15) MPa,抗弯曲强度为(120±9) MPa,均达到或超过PMMA骨水泥的力学性能。 由于磷酸钙骨水泥的力学强度较PMMA低,为改善力学强度,Vlad等[9]将α-Fe2O3纳米颗粒掺入磷酸钙骨水泥中,发现不仅可提高磷酸钙骨水泥的抗压强度、可注射性、延长凝固时间,而且具有良好的生物相容性;但磁性纳米颗粒的掺入量并非与骨水泥的力学强度呈正比,含质量百分比8%纳米颗粒磷酸钙骨水泥的抗压强度较单纯磷酸钙骨水泥强,而质量百分比24%纳米颗粒磷酸钙骨水泥的抗压强度较含质量百分比8%纳米颗粒的磷酸钙骨水泥弱,均小于70 MPa,原因是由于磁性纳米颗粒所占质量百分比过高时,与磷酸钙粉末混合不均匀,凝固后会在磁性纳米颗粒含量高的区域形成力学薄弱区,从而导致强度下降。Kawashita等[10]将不同质量百分比(40%,50%,60%)50-500 nm的Fe3O4磁性纳米颗粒与PMMA混合,分别制成M-40C、M-50C、M-60C磁性骨水泥,并与单纯PMMA进行对照研究,随着磁性纳米颗粒含量增加,骨水泥的凝固时间逐渐延长,但抗压强度差异不大,M-40C磁性骨水泥的抗压强度为(91.4±6.1) MPa,M-50C磁性骨水泥的抗压强度为(89.2±6.5) MPa,而M-60C磁性骨水泥难以成形,无法测量骨水泥的抗压强度和凝固温度。早期骨水泥掺入的磁响应物质含量均较高,质量百分比最高达到50%。近年来,重庆医科大学研究团队向PMMA与磷酸钙骨水泥2种骨水泥基质中掺入含质量百分比10%的Fe3O4纳米颗粒,合成磁性骨水泥[11-12],发现其具有良好的体内、外消融作用,但未行力学评价。Vlad等[9]制作的含质量百分比8%磁性纳米颗粒的磁性磷酸钙骨水泥,颗粒含量虽低,但目的在于增强磷酸钙骨水泥的力学强度和可注射性,并非用于热疗,究其原因是颗粒的产热性能差,需要较高质量百分比的磁性纳米颗粒,才能在交变磁场中达到热疗所需的温度。刘义灏等[13]以PMMA为基质,掺入质量百分比5%的Co0.04Fe2.96O4纳米颗粒,其抗压强度为(77.5±6.1) MPa,达到了ISO5833标准,在 390 kHz、22 kA/m的交变磁场中,3 min可达到50 ℃,满足临床热疗所需温度。 综上,少量掺入磁性纳米颗粒会增加磁性骨水泥的力学强度,但掺入量过多反而导致其力学强度的下降,过多的掺入(60%)甚至会引起骨水泥难以凝固。骨水泥凝固时间与颗粒掺入量呈正比[10],虽然凝固时间的延长,便于临床操作,但以磷酸钙为基质的磁性骨水泥本身凝固时间较长,凝固时间的进一步延长反而增加骨水泥渗漏的风险,含量过高甚至会导致颗粒没有被完全固化而出现洗脱现象,增加生物毒性[11]。聚合温度会随着磁性纳米颗粒含量的增加而呈下降趋势,可能与单位质量内单体体积的下降有关[10]。可注射性与磁性纳米颗粒含量呈正比[12],但同样有增加骨水泥渗漏的风险。有关磁性骨水泥的文献,见表1。"

2.2 磁性骨水泥的体外实验 2.2.1 生物相容性 生物相容性是医用材料评价的重要指标,磁性骨水泥生物相容性评价的对象是磁性骨水泥浸提液,可参照ISO10993.12-2009标准制备骨水泥浸提液[14]。Li等[15]采用DNA定量法评价含质量百分比30%Fe3O4纳米颗粒磁性PMMA骨水泥的生物相容性,发现孵育1,4,7 d 磁性骨水泥的生物相容性与对照组(空白PMMA)比较差异无统计学意义。唐正海等[16]将Fe3O4磁性纳米颗粒分别与PMMA和磷酸钙骨水泥混合,制成不同质量百分比(10%,20%)的磁性骨水泥,研究发现随着磁性纳米颗粒比例的增高,骨水泥的毒性逐渐增加,磁性PMMA骨水泥具有轻、中度细胞毒性,而磁性磷酸钙骨水泥生物相容性好,基本无细胞毒性。刘义灏等[13]制备了含质量百分比5%C0.04Fe2.96O4纳米颗粒的磁性PMMA,发现其生物毒性与单纯PMMA比较差异无统计学意义。Verné等[17]与Miola等[18]将磁性生物活性陶瓷粉(SC45)与PMMA按照不同质量百分比(10%,15%,20%)混合,制成磁性骨水泥,将其浸提液与人骨肉瘤细胞MG63细胞共孵育24,48,72 h,MTT法检测细胞毒性发现,含质量百分比10%,15%,20%磁性生物活性陶瓷粉骨水泥各时间点的细胞增殖与对照组比较无明显差异。 综上,磁性骨水泥浸提液的毒性与骨水泥基质、掺杂磁性纳米颗粒的含量密切相关,以PMMA为基质的磁性骨水泥细胞毒性要高于磷酸钙骨水泥,而掺杂的磁性纳米颗粒含量越高,骨水泥的生物相容性越差,降低磁性纳米颗粒的掺杂量,是降低骨水泥毒性、提高其生物相容性的重要途径。 2.2.2 热疗作用 热疗作用是磁性骨水泥研究的核心,高产热效能的磁性纳米颗粒,可减少颗粒质量百分比,便于量产,能更好地满足临床需求。磁性纳米颗粒在交变磁场中的产热能力通常用比吸收率来表征,是指单位质量磁性纳米颗粒产热的能力,单位是Wg-1,比吸收率与颗粒的成分、尺寸、形貌相关,会随着磁场的频率和磁场强度的增大而增加。超顺磁性纳米颗粒通过奈尔弛豫和布朗弛豫产热[19],奈尔弛豫与颗粒的自身特征,如颗粒成分、尺寸、形貌有关,布朗弛豫与颗粒所在溶质的黏稠度有关,生物体应用中布朗弛豫常被阻断,主要依靠奈尔弛豫产热,产热能力会降低[20]。骨水泥固化后,磁性纳米颗粒的体转动消失,布朗弛豫被阻断,产热能力也会降低,而为患者安全起见,磁场强度和频率的乘积需小于5×109 Am-1s-1[21],而且肿瘤局部血液循环丰富,会出现温度衰减现象,因此,用于合成磁性骨水泥的磁性纳米颗粒必须具有高的产热效能,才能满足生物应用的需求。 Portela等[22]研制的掺杂磁性生物陶瓷粉磁性骨水泥,其比吸收率值最高仅为2.11 Wg-1,难以满足临床热疗的要求。Takegami等[8]发现在800 e、100 kHz的交变磁场中,含质量百分比80%微米级Fe3O4磁性骨水泥的最高温度为61 ℃。Kawashita等[10]发现在频率为 100 kHz的磁场中,含质量百分比40%,50%磁性纳米颗粒的磷酸钙骨水泥,当场强为3 000 e时能在几十秒之内温度超过70 ℃;当场强为1 200 e时,在500 s之内分别可达到40,48 ℃。Li等[15]研究加入不同粒径(300,35,11 nm)磁性纳米颗粒PMMA骨水泥的理化性能,指出磁性骨水泥的产热能力与粒径大小及所用磁场的条件密切相关。Guardia等[23]研究发现,(19±3) nm的超顺磁性纳米颗粒比吸收率值高达2 452 Wg-1。韩国研究小组Lee等[24]报道了具有核壳结构的交换耦合磁性纳米颗粒,其在交变磁场中的比吸收率达到3 886 Wg-1。贺淑莉[25]研究团队发现软磁和硬磁元素掺杂的铁氧体纳米颗粒,可提高颗粒的比吸收率值,22 nm的Co0.03Mn0.28Fe2.7O4/SiO2在交变磁场中的比吸收率值可达到3 417 Wg-1,以PMMA为基质的磁性骨水泥,只需添加质量百分比1%的磁性纳米颗粒即可满足临床热疗的要求[26],见图2,3。 2.3 体内实验 2.3.1 动物模型 磁性骨水泥研究多采用免疫缺陷动物移植肿瘤模型,采用裸鼠皮下注射肿瘤细胞悬液建立动物模型[27],虽然具有较高的成瘤率、位置表浅、便于观察等优点,但用于磁性骨水泥研究,可能存在以下问题:①骨水泥主要用于骨肿瘤的治疗,接种于皮下的肿瘤属软组织肿瘤,其热疗时热传导及生物学特征与骨肿瘤均存在很大差异,如采用骨髓腔内注射造模[28],由于裸鼠体型小,又不便于骨水泥髓腔内注射操作;②磁热疗的优势在可运用于较深部位的肿瘤[29],而将肿瘤接种于皮下,难以体现磁热疗的优势。日本三重大学研究团队采用VX2移植骨转移瘤模型作为磁性骨水泥磁热疗研究模型[30],VX2细胞是人类乳头瘤病毒诱发的鳞状细胞癌,新西兰兔、日本大耳白兔等先天对其缺乏免疫,其具有成瘤率高、传代稳定、早期容易出现肺转移等特征,与临床原发性骨肿瘤(骨肉瘤)及骨转移瘤等生物学特征具有相似之处,是研究磁性骨水泥较为理想的动物模型。 2.3.2 磁热疗方案 磁热疗的时间、频次、疗程及温度文献报道存在很大差异。Kusaka等[31]制作了兔VX2移植骨肿瘤模型,成模1周后在兔胫骨髓腔内注射磁性骨水泥,治疗50 min,仅治疗1次。Matsumine等[30]对患者在磁性磷酸钙骨水泥注射1周后,于术后8,10,12,15,17,19,22,24,26,29 d进行磁热疗,每次治疗15 min,3例患者采用有创光纤测温,骨皮质温度最高控制在42 ℃。Portela等[32]制备了C57BL6恶性黑色素瘤动物模型,在磁性骨水泥注射后每次治疗30 min,间隔48 h治疗1次,肿瘤局部温度在5 min内升至38- 39 ℃后维持30 min,但骨水泥的温升效果欠佳,未达到热疗所需的43-45 ℃。Xu等[12]将注射磁性磷酸钙骨水泥的裸鼠肿瘤模型置于线圈中180 s,并用红外相机检测温度变化,肿瘤局部温度达到(74.73±9.12) ℃。Ling等[11]将注射磁性PMMA骨水泥的裸鼠肿瘤模型置于线圈中180 s,肿瘤局部温度达到(53.05±3.18) ℃。 综上可见,磁热疗的方案存在很大差异,温度变化范围与所用磁性骨水泥的升温性能有关,但热疗指温度在41-46 ℃之间,而超过56 ℃即为热消融,而且两者治疗后的病理生理也不相同,热疗后肿瘤细胞以凋亡为主,而热消融后组织以坏死为主,组织坏死容易导致局部炎症细胞浸润,甚至出现红肿热痛等临床表现[32]。因此,热疗被更多的学者推崇,故而磁热疗过程中温度不宜过高,控制43-45 ℃之间为宜。 2.3.3 测温方法 测温是热疗的关键技术,也是确保热疗安全进行的措施,热疗的温度需控制在43-45 ℃之间,温度过高易造成周围正常组织被灼伤。目前测温方法主要有实时光纤测温和红外成像技术,这2种方法适合于表浅的肿瘤,而对于深部肿瘤,如骨肿瘤、消化道肿瘤等难以实现温度监测。虽然日本三重大学研究团队采用多通道光纤实时测温系统,对VX2移植肿瘤模型骨水泥注射的不同部位温度变化进行了间接测温[30],但同样会对动物造成损伤;而Oliveira等[33]将光纤插入膀胱内监测腔内温度变化,操作过程均较为繁琐,且有造成动物膀胱损伤的风险。红外成像技术可实施、准确的检测温度,在光热疗过程中尤为实用[34],但仅限于观察动物体表的温度变化,对于较深部位的骨肿瘤模型难以实施温度监控。因此,准确、实时、无创测温,将会是磁热疗面临的技术难题。热籽与肿瘤组织充分接触,是杀灭肿瘤细胞的前提,磁性骨水泥在磁场中以磁性骨水泥的中心温度最高,热衰减的程度与距离呈负相关。Takegami等[8]通过多通道光纤测温发现,注入兔胫骨骨髓腔内的骨水泥中心温度达到50-60 ℃时,骨皮质表面的温度会升高至43-45 ℃,但在髓腔内,距离骨水泥1 cm以上时温度难以超过40 ℃,而采用尸体胫骨对温度在骨中传导的研究发现,所得结果与动物实验相似,但在尸体骨中升温速率更快,可能与骨水泥的体积较大有关。 2.3.4 评价指标 肿瘤模型采用的评价指标主要有肿瘤的体积、质量,影像学包括B超、X射线、micro-CT和核磁,血液检测指标包括肿瘤免疫指标、生化指标等,病理学包括苏木精-伊红染色,细胞凋亡TUNEL染色、免疫组织化学等。不同的动物模型需选择不同的评价指标,如肿瘤的体积、质量及B超,不适用于VX2移植骨肿瘤模型,需采用患肢肿瘤周径来评价肿瘤的进展过 程[31];而对于皮下移植的肿瘤模型,可实时动态观察肿瘤的体积,在治疗结束时可完整获得肿瘤的质量进行评价。X射线、micro-CT可评估骨质破坏及骨小梁结构变化[28],而核磁在发现骨肿瘤是否成模方面的敏感性和特异性均较高[35],适用于早期诊断骨肿瘤。肿瘤免疫治疗主要用于观察磁性骨水泥磁热疗后血液免疫指标的变化[36],而生化指标主要为肝、肾功能,用于评价磁性骨水泥的体内毒性[37]。病理学苏木精-伊红染色可观察肿瘤组织热疗后的形态改变,包括坏死、凋亡、纤维化等,TUNEL染色可观察与磁性骨水泥、肿瘤组织界面不同距离细胞的凋亡指数[38],配合免疫组织化学染色,可进一步明确磁热疗对肿瘤血管生成的影响作用。 "

| [1]Owari T,Miyake M,Nakai Y,et al.Clinical Features and Risk Factors of Skeletal-Related Events in Genitourinary Cancer Patients with Bone Metastasis: A Retrospective Analysis of Prostate Cancer, Renal Cell Carcinoma, and Urothelial Carcinoma. Oncology.2018;95(3):170-178.[2]Li S,Peng Y,Weinhandl ED,et al.Estimated number of prevalent cases of metastatic bone disease in the US adult population. Clin Epidemiol. 2012; 4:87-93.[3]Yuasa T,Urakami S.Kidney cancer: decreased incidence of skeletal-related events in mRCC.Nat Rev Urol. 2014;11(4): 193-194.[4]Galibert P,Deramond H,Rosat P,et al.Preliminary note on the treatment of vertebral angioma by percutaneous acrylic vertebroplasty. Neurochirurgie.1987;33:166-168.[5]田庆华,吴春根,顾一峰,等.经皮骨成形术治疗椎外转移性骨肿瘤的应用[J].介入放射学,2012,21(8):645-650.[6]Kim YI, Kang HG, Kim JH,et al.Closed intramedullary nailing with percutaneous cement augmentation for long bone metastases.J Bone Joint J.2016;98(5):703-709.[7]Gilchrist RK,Medal R,Shorey WD,et al.Selective inductive heating of lymph nodes.Ann Surg. 1957;146(4): 596-606.[8]Takegami K,Sano T,Wakabayashi H,et al. New ferromagnetic bone cement for local hyperthermia.J Biomed Mater Res.1998; 43(2):210-214. [9]Vlad MD,del Valle LJ,Barracó M,et al.Iron Oxide Nanoparticles Significantly Enhances the Injectability of Apatitic Bone Cement for Vertebroplasty.Spine(Phila Pa 1976).2008;33(21): 2290-2298.[10]Kawashita M,Kawamura K,Li Z.PMMA-based bone cements containing magnetite particles for the hyperthermia of cancer.Acta Biomater. 2010; 6(8):3187-3192.[11]Ling Y,Tang X,Wang F,et al.Highly efficient magnetic hyperthermia ablation of tumors using injectable polymethylmethacrylate–Fe3O4.RSC Adv.2017;7(5): 2913-2918. [12]Xu C,Zheng Y,Gao W,et al.Magnetic Hyperthermia Ablation of Tumors Using Injectable Fe3O4/Calcium Phosphate Cement.ACS Appl Mater Interfaces.2015;7(25):13866-13875.[13]刘义灏,俞翔,张力,等.一种新型磁性骨水泥的制备及其在交变磁场中对骨肉瘤MG-63细胞凋亡的影响[J].江苏大学学报(医学版), 2017,27(1):5-9.[14]Frassica DA,Frassica FJ.Nonoperative management. In:Simon MA, Springfield D (eds) .Surgery for bone and soft-tissue tumors. Lippincott-Raven,Philadelphia,1998: 633-637. [15]Li Z,Kawamura K,Kawashita M,et al.In vitro assessment of poly(methylmethacrylate)-based bone cement containing magnetite nanoparticles for hyperthermia treatment of bone tumor. J Biomed Mater Res Part A.2012;100A:2537-2545.[16]唐正海,王晓文,张友仁,等.磁性复合骨水泥的体外细胞毒性[J].中国组织工程研究,2013,17(21):3937-3943. [17]Verné E,Bruno M, Miola M,et al.Composite bone cements loaded with a bioactive and ferrimagnetic glass-ceramic: Leaching, bioactivity and cytocompatibility.Mater Sci Eng C Mater Biol Appl. 2015;53:95-103.[18]Miola M, Laviano F,Gerbaldo R,et al.Composite bone cements for hyperthermia:modeling and characterization of magnetic, calorimetric and in vitro heating properties.Ceram Int. 2017;43(6):4831-4840.[19]Blanco-Andujar C,Walter A,Cotin G,et al.Design of iron oxide-based nanoparticles for MRI and magnetic hyperthermia.Nanomedicine. 2016; 11(14):1889-1910.[20]Di Corato R,Espinosa A,Lartigue L,et al.Magnetic hyperthermia efficiency in the cellular environment for differentnanoparticle designs.Biomaterials. 2014;35(24): 6400-6411.[21]Hergt R,Dutz S.Magnetic particle hyperthermia-biophysical limitations of a visionary tumour therapy.J Magn Magn Mater.2007;311(1):187-192.[22]Portela A,Vasconcelos M,Branco R,et al.An in vitro and in vivo investigation of the biological behavior of a ferrimagnetic cement for highly focalized thermotherapy.J Mater Sci Mater Med.2010;21(8): 2413-2423.[23]Guardia P,Di Corato R,Lartigue L,et al. Water-soluble iron oxide nanocubes with high values of specific absorption rate for cancer cell hyperthermia treatment.ACS Nano.2012;6(4): 3080-3091. [24]Lee JH,Jang JT,Choi JS,et al. Exchange-coupled magnetic nanoparticles for efficient heat induction.Nat Nanotechnol. 2011;6(7):418-422.[25]He S,Zhang H,Liu Y,et al.Maximizing Specific Loss Power for Magnetic Hyperthermia by Hard-Soft Mixed Ferrites.Small. 2018:e1800135. [26]贺淑莉,刘义灏,俞翔,等.一种具有高产热效率的骨水泥[P].北京: CN106267330A,2017-01-04.[27]Tang X,Xu Y,Chen J,et al.Intermittent time-set technique controlling the temperature of magnetic-hyperthermia- ablation for tumor therapy.RSC Adv.2018;8(30):16410-16418. [28]Wang Y,Yang J,Liu H, et al.Osteotropic peptide-mediated bone targeting for photothermal treatment of bone tumors. Biomaterials.2017;114: 97-105.[29]Derfus AM,von Maltzahn G,Harris TJ,et al. Remotely triggered release from magneticnanoparticles.Adv Mater.2007;19(22): 3932-3936.[30]Matsumine A,Takegami K,Asanuma K,et al.A novel hyperthermia treatment for bone metastases using magnetic materials.Int J Clin Oncol. 2011;16(2):101-108. [31]Kusaka M,Takegami K,Sudo A,et al.Effect of hyperthermia by magnetite cement on tumor-induced bone destruction.J Orthop Sci.2002;7(3): 354-357. [32]Portela A,Vasconcelos M,Fernandes MH,et al.Highly focalised thermotherapy using a ferrimagnetic cement in the treatment of a melanoma mouse model by low temperature hyperthermia. Int J Hyperthermia.2013;29(2):121-132.[33]Oliveira TR,Stauffer PR,Lee CT,et al.Magnetic fluid hyperthermia for bladder cancer: A preclinical dosimetry study. Int J Hyperthermia. 2013; 29(8):835-844. [34]He C,Duan X,Guo N,et al.Core-shell nanoscale coordination polymers combine chemotherapy and photodynamic therapy to potentiate checkpoint blockade cancer immunotherapy.Nat Commun. 2016;7: 12499.[35]Melancon MP,Appleton Figueira T,Fuentes DT,et al. Development of an Electroporation and Nanoparticle-based Therapeutic Platform for Bone Metastases.Radiology. 2018;286(1):149-157. [36]Chen Q,Xu L,Liang C,et al.Photothermal therapy with immune-adjuvant nanoparticles together with checkpoint blockade for effective cancer immunotherapy.Nat Commun. 2016;7:13193. [37]Shetake NG, Kumar A,Gaikwad S,et al.Magnetic nanoparticle-mediated hyperthermia therapy induces tumour growth inhibition by apoptosis and Hsp90/AKT modulation.Int J Hyperthermia.2012;31(8):909-919. [38]Xie J,Zhang Y,Yan C,et al.High-performance PEGylated Mn-Zn ferrite nanocrystals as a passive-targeted agent for magnetically induced cancer theranostics.Biomaterials. 2014; 35(33):9126-9136. [39]Datta NR,Krishnan S,Speiser DE,et al.Magnetic nanoparticle-induced hyperthermia with appropriate payloads: Paul Ehrlich's "magic (nano)bullet" for cancer theranostics? Cancer Treat Rev.2016;50: 217-227. |
| [1] | Chen Xinmin, Li Wenbiao, Xiong Kaikai, Xiong Xiaoyan, Zheng Liqin, Li Musheng, Zheng Yongze, Lin Ziling. Type A3.3 femoral intertrochanteric fracture with augmented proximal femoral nail anti-rotation in the elderly: finite element analysis of the optimal amount of bone cement [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1404-1409. |
| [2] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [3] | Yuan Jun, Yang Jiafu. Hemostatic effect of topical tranexamic acid infiltration in cementless total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 873-877. |
| [4] | Hou Guangyuan, Zhang Jixue, Zhang Zhijun, Meng Xianghui, Duan Wen, Gao Weilu. Bone cement pedicle screw fixation and fusion in the treatment of degenerative spinal disease with osteoporosis: one-year follow-up [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 878-883. |
| [5] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [6] | Zhong Yuanming, Wan Tong, Zhong Xifeng, Wu Zhuotan, He Bingkun, Wu Sixian. Meta-analysis of the efficacy and safety of percutaneous curved vertebroplasty and unilateral pedicle approach percutaneous vertebroplasty in the treatment of osteoporotic vertebral compression fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 456-462. |
| [7] | Meng Lingjie, Qian Hui, Sheng Xiaolei, Lu Jianfeng, Huang Jianping, Qi Liangang, Liu Zongbao. Application of three-dimensional printing technology combined with bone cement in minimally invasive treatment of the collapsed Sanders III type of calcaneal fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3784-3789. |
| [8] | Feng Guancheng, Fang Jianming, Lü Haoran, Zhang Dongsheng, Wei Jiadong, Yu Bingbing. How does bone cement dispersion affect the early outcome of percutaneous vertebroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3450-3457. |
| [9] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [10] | Sun Yiqiang, Xing Jianqiang, Li Xuecheng, Wang Xin, Tian Shenglan, Zhao Zihao, Geng Xiaopeng. Kyphoplasty versus vertebroplasty in the treatment of osteoporotic vertebral compression fracture in the elderly: a comparison of vertebral height recovery [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(18): 2851-2855. |
| [11] | Li Qiang, Li Jun, Luan Jian, Jin Canghai, Hao Meng, Lin Yong. Bone cement distribution of percutaneous curved vertebroplasty for the treatment of osteoporotic vertebral compression fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2466-2471. |
| [12] | Tian Lin, Shi Xiaoqing, Mao Jun, Zhang Nongshan, Zhang Li, Xing Runlin, Wang Peimin. Meta-analysis of vacuum-sealing drainage combined with antibiotic bone cement in the treatment of chronic osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2618-2624. |
| [13] | Song Min, Lu Chao, Chen Jin, Wu Gaoyi, Li Congcong, Li Anan, Ye Guozhu, Lin Wenzheng, Cai Yuning, Liu Wengang, Xu Weipeng. Application of tourniquet affects thickness of bone cement penetration in total knee arthroplasty: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(12): 1917-1923. |
| [14] | Zhong Yuanming, Wan Tong, Zhong Xifeng, Wu Zhuotan, He Bingkun, Wu Sixian. Meta-analysis of efficacy and safety of bone filling bag vertebroplasty and percutaneous vertebroplasty in the treatment of osteoporotic vertebral compression fracture#br# [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(12): 1930-1935. |
| [15] | He Renjian, Yu Chao, Luo Yuanchao, Liu Xu, Yang Fuguo. Safety dose of bone cement in vertebroplasty assessed by Mimics software [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1482-1488. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||