Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (29): 4743-4748.doi: 10.3969/j.issn.2095-4344.0635
Previous Articles Next Articles
Stem cells for ophthalmic diseases: recovery from damage and functional reconstruction
Zhou Li1, Tian Bin2, Ji Yuan-hong2, Zhou Zheng3, Wei Xiao-dan3
- 1Department of Ophthalmology, 2Department of Rehabilitation, Rehabilitation Hospital, Tangshan Gongren Hospital Group, Tangshan 063000, Hebei Province, China; 3Department of Ophthalmology, Tangshan Gongren Hospital, Tangshan 063000, Hebei Province, China
-
Revised:2018-06-04Online:2018-10-18Published:2018-10-18 -
About author:Zhou Li, Attending physician, Department of Ophthalmology, Rehabilitation Hospital, Tangshan Gongren Hospital Group, Tangshan 063000, Hebei Province, China
CLC Number:
Cite this article
Zhou Li, Tian Bin, Ji Yuan-hong, Zhou Zheng, Wei Xiao-dan. Stem cells for ophthalmic diseases: recovery from damage and functional reconstruction[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(29): 4743-4748.
share this article

2.1 干细胞的特性 干细胞的主要特征是可以复制出和亲代完全相同基因型的细胞,干细胞可根据其分化潜能和来源不同分为胚胎干细胞和成体干细胞两类[3-4]。胚胎干细胞是一种具有胚胎属性,可以向不同胚层分化的全能干细胞,具有形成完整个体的分化潜能,但是其取材和应用受伦理和社会等方面的限制。成体干细胞来自许多不同类型的细胞,主要包括骨髓间充质干细胞(bone marrow mesenchymal stem cells,BMSCs)、脂肪间充质干细胞(adipose tissue-derived stromal cells,ADSCs)、脐带间充质干细胞(umbilical cord mesenchymal stem cells,UMSCs)、胎盘间充质干细胞(placenta-derived mesenchymal stem cells,PMSCs)、脐血间充质干细胞(umbilical cord blood mesenchemal stem cells,UCBMSCs)、神经干细胞(neural stem cells,NSCs)、角膜缘干细胞(limbalStem cells,LSCs)、表皮干细胞(epidermal stem cells,ESCs)和牙髓干细胞(dental pulp stem cells,DPSCs)等[5]。这些细胞来源于胚胎发育早期的中胚层和外胚层,由于成体干细胞已经失去了胚胎时期的一些生物学特征,因此这种细胞虽然也具有向不同胚层分化的功能但是分化能力比胚胎干细胞弱,但其来源广泛,免疫原性低、安全性好、无伦理学限制,目前在临床上已经得到了广泛的应用。 2.2 干细胞在眼科疾病中的应用 2.2.1 干细胞在眼表疾病中的应用 Steven-Jonson综合征、热学或化学性损伤、类天疱疮等均可引起角膜浑浊和视觉的严重损伤。这些疾病发病特点为患病率高、致盲性极强且不易治愈。眼表疾病的发病机制主要是具有修复能力的角膜缘干细胞群损伤,致使干细胞缺失和角膜“结膜化”,从而导致一系列的炎性反应。传统的自体或者同种异体角膜移植治疗效果不满意,主要由于供体来源不足及移植排斥反应等原因。因此,干细胞的研究为解决这一难题带来希望。 (1)胚胎干细胞在眼表疾病中的应用:1998年,Thomson等[6]在IVF实验室中,利用废弃的胚胎第1次培养出了胚胎干细胞,推动人们对胚胎干细胞的研究热潮。目前,胚胎干细胞与眼角疾病的研究主要有3个方向:第1个方向是按照起源说,模拟胚体的方法,经过培养后结合于特定的细胞外基质,定向为角膜缘干细胞。Hanson等[7]研究中提及将GFP标记的人胚胎干细胞在DEF-CS培养体系中预分化22 d,然后将预分化的人胚胎干细胞移植到人角膜基质(保留Bowman膜)共培养,9 d后移植细胞形成1-4层角膜类上皮,并出现角膜特异性标志物如PAX6和K3;第2个方向是模拟角膜缘干细胞龛的过程,将角膜缘干细胞通过定向诱导的方式完成角膜上皮的分化,并在角膜重建中发挥作用。2014年Zhang等[8]研究中采用一定的材料作为基质,如羊膜或者Ⅳ型胶原,将胚胎干细胞与人角膜缘成纤维细胞混合共培养,进行定向诱导分化,分化后的细胞表达特异性的标识K3,而不表达胚胎干细胞的标记OCT4;第3个方向是基因改造。在胚胎早期,基因Pax6对胚胎的视器分化起重要的作用。Kayama等[9]将pax6/gfp/neoR与猕猴胚胎干细胞混合培养14 d后移植到损伤的角膜,结果证实这种方法能够获得特异性好、纯度高的角膜上皮。胚胎干细胞可以为眼表修复提供充足的移植细胞来源,既可在体外增殖,又能维持不分化状态,从而解决这一眼科难题(表1)。遗憾的是胚胎干细胞取材和应用受伦理和社会等方面的限制。"

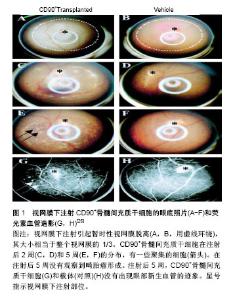
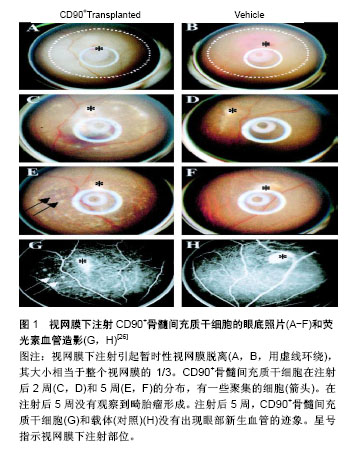
(2)成体干细胞在眼表疾病中的应用:骨髓间充质干细胞是一种存在骨髓中的非造血细胞,在适当的诱导条件下可分化为多种组织细胞,其取材方便不受伦理的限制,而且还可诱导免疫耐受,是目前组织工程的研究热点。刘德伍等[10]以角膜基质作为支架材料,将骨髓间充质干细胞培养于此基质上,能够定向诱导分化为角膜细胞,成功构建组织工程化人工活性角膜,此研究可有望成为一种修复角膜损伤的新方法。Ma等[11]将人骨髓间充质干细胞移植到化学烧伤的小鼠眼角膜,研究结果证实经羊膜扩增后移植的骨髓间充质干细胞可对角膜损伤有修复作用。2017年孙红等[12]研究了骨髓间充质干细胞对碱烧伤兔角膜修复的促进机制,得出结论:静脉输入骨髓间充质干细胞能够加快兔角膜的修复速度,在此基础上兔角膜的透明度得到提高,其机制可能与角膜血管生成的精细调控有关。 众所周知,皮肤在人体中占面积最多,所以皮肤干细胞有着其独特的优势:一是取材方便;二是排除了免疫功能上的排斥反应。皮肤干细胞在眼科有着不可估量的研究和应用价值。目前研究显示皮肤干细胞和毛囊干细胞在眼科领域中的应用较为广泛。2014年7月,在《Nature》杂志发表的一篇文章“WNT7A and PAX6 definecorneal epithelium homeostasiand pathogenesis”,该研究是由中山大学眼科学团队与美国研究人员合作的成果,该研究证实了调控因子WNT7A和PAX6在角膜缘干细胞分化中起着重要的作用,对于角膜表面的修复与再生和治疗角膜缘干细胞缺陷患者提供了一个可以期望的前景[13]。Tzameret等[14]研究在以Ⅳ型胶原为基质的基础上,通过 integrina6 抗体分选鼠须毛囊干细胞,分选后的细胞表达角膜上皮的特异性标记物K12和PAX6,不表达K10。Yang等[15]研究表明角膜上皮被皮肤干细胞重建后,已经不表达皮肤的标志物,仅表达角膜的特异性标记物K3/K12和PAX6,且角膜重建效果良好,无并发症。在2010年,意大利研究人员选取112例被化学灼伤而导致失明的患者,用患者自身角膜缘干细胞治疗角膜损伤[16]。该研究在患者的眼白和角膜间的异组织边缘提取干细胞,并在体外进行诱导培养使其发育为纤维组织,然后植入患者的眼睛受损部位。有些患者角膜缘完全缺失的情况下,移植角膜缘干细胞与羊膜复合物,仍能产生一定的疗效[17]。 诱导多潜能干细胞的主要特点表现在基因型、端粒酶及生物标记等,其各项特征均与胚胎干细胞相似。国外多项研究表明,诱导多潜能干细胞在向视器分化过程中受多种因素调控[18-20]。Pax6基因与先天性眼表疾病密切相关,其突变可导致眼病发生,如无虹膜症。国外有学者等研究发现miR-450-5p可抑制角膜上皮标记物Pax6的表达,如果敲除外胚层的miR-450-5p基因,则Pax6表达不被抑制,从而分化为角膜上皮[21]。国内学者在Ⅳ型胶原铺板的 Transwell 体系中将鼠角膜缘基质细胞与传代鼠诱导多潜能干细胞共培养,共培养2周以后获得角膜类上皮细胞,其细胞表达标记物Nanog、Pax67、P63、K12,不表达SSEA3;分化后细胞仍表达诱导多潜能干细胞的特异性标记物Pax6和P6,是分化不彻底的标志,根据实验结果计算得出表达角膜上皮特异性标记物K12的细胞仅仅占13%[22]。另有学者利用骨形态发生蛋白4作为诱导分子,将全角膜基质细胞作为诱导材料成功将角膜上皮的分化率提高,可以根据这些研究得出结论,骨形态发生蛋白4在促进角膜分化的过程中起重要作用[23]。尽管诱导多潜能干细胞在眼科领域的应用很广泛,但是它的分化率和安全性等方面仍存在不可忽视的问题。 2.2.2 干细胞在视网膜疾病中的应用 视网膜中有一种色素上皮细胞,呈多角形,排列十分规则。色素上皮细胞ZO-1,bestrophin,MiTF等标志物呈阳性,OCT4,NANOG,SOX2等标志物呈阴性[24]。 (1)胚胎干细胞在视网膜疾病中的应用:随着中国人口老龄化的加快,糖尿病视网膜病变、视网膜色素增生、视网膜黄斑性病变等眼科疾病逐年增长。视网膜退行性疾病的病理机制是感光细胞、神经节细胞、色素上皮细胞等进行性变性、凋亡、坏死,引起视网膜各级神经元的结构和功能异常,最终造成不可逆性的视功能损害。目前干细胞技术已被广泛应用于眼科疾病的治疗中,并且其应用效果更是获得广大眼科疾病患者及医学工作者的认可。 胚胎干细胞是一种全能干细胞,无论是在体内还是在体外培养都能被诱导分化为机体几乎所有的细胞类型。徐国彤教授曾在2016年第四届国际实验生物学和医学论坛分享了关于干细胞治疗视网膜变性疾病的研究进展,提出胚胎干细胞和诱导多能干细胞治疗视网膜疾病的临床应用,以完全分化为成熟的视网膜色素上皮细胞来达到治疗目的,这对于角膜发生严重混浊需要移植的患者,或患有黄斑变性等视网膜疾病的患者,甚至对青光眼患者,都带来了新的希望。 在眼科疾病的治疗中,将胚胎干细胞定向诱导分化后移植,这项研究目前仅在动物体内得以实现。研究表明胚胎干细胞经特定的诱导能够分化为类视网膜神经节细胞。除此之外另有研究表明,视黄醇能够定向诱导胚胎干细胞分化为视网膜感光细胞,将其移植入缺血再灌注的视网膜下间隙可起到很好的效果。国外学者Haruta等[25]以皇家外科学院(Royal College of surgeon,RCS)鼠的视网膜为实验对象,将来源于胚胎干细胞的色素上皮细胞移植到RCS鼠正常饲养8周,各项数据证实由胚胎干细胞诱导分化后的色素上皮细胞表达所有特异性的标记,为研究视网膜变性的发病机制及调控途径提供了新思路。成瘤性是胚胎干细胞的一个不可避免的缺点,这就要求在应用它之前必须完成两件事:首先是定向将胚胎干细胞分化为某种组织;其次是设计能够调控自我凋亡的基因-自杀基因。自杀基因的任务就是体内移植的细胞变异为肿瘤的时候,能够立即启动凋亡途径,以消灭肿瘤。各位学者目前正在研究如何控制胚胎干细胞在分裂增殖过程中不发生突变,产生正常分化的细胞。 目前,已在许多小鼠模型及人体试验中开展视网膜上皮细胞移植技术[26-27]。Lamba等[28]通过实验已经证实胚胎干细胞可被直接诱导分化为视网膜细胞及视网膜上皮细胞,且诱导后的细胞移植人体内能够被整合进视网膜,并表达光感受器细胞标记。另外一学者研究表明在体外成功诱导胚胎干细胞高效分化为多角形视网膜上皮细胞,同时向培养液中添加碱性成纤维细胞生长因子、牛磺酸和维生素A酸,产生大量的视锥细胞和视杆细胞[29]。此研究为治疗视网膜变性疾病(如黄斑变性或色素性视网膜炎)提供了一种可能的途径。 (2)成体干细胞在视网膜疾病中的应用:骨髓间充质干细胞在体外可以诱导分化为视网膜样细胞,具有治疗视网膜退行性病变的潜力。Kicic等[30]利用活化蛋白A、牛磺酸和表皮生长因子在体外成功将人CD90+骨髓间充质干细胞诱导分化为光感受器样细胞,表达光感受器细胞特异性标志物视紫红质、视蛋白、视觉恢复蛋白;将CD90+骨髓间充质干细胞注射到成年大鼠视网膜下腔2周,其可整合人宿主视网膜,形成结构类似于感光细胞样细胞并表达感光细胞特异性标记物,而未见畸胎瘤的形成(图1)。"
| [1] 王延华.干细胞理论与技术[M].北京:科学出版社,2005.[2] 张元中.干细胞临床治疗——希望与挑战[J].感染、炎症、修复, 2010,11(3):132.[3] Nazari H, Zhang L, Zhu D, et al. Stem cell based therapies for age-related macular degeneration: The promises and the challenges. Prog Retin Eye Res. 2015;48:1-39.[4] Thomson JA, Itskovitz-Eldor J, Shapiro SS, et al. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282(5391):1145-1147.[5] Takahashi M, Sato T, Kishi S. Intravitreal bevacizumab for age-related macular degeneration with good visual acuity. Jpn J Ophthalmol. 2010;54(6):565-570.[6] Thomson JA, Burkholder DA, Heithaus MR, et al. Extreme temperatures, foundation species, and abrupt ecosystem change: an example from an iconic seagrass ecosystem. Glob Chang Biol. 2015;21(4):1463-1474.[7] Hanson C, Hardarson T, Ellerström C, et al. Transplantation of human embryonic stem cells onto a partially wounded human cornea in vitro. Acta Ophthalmol. 2013;91(2):127-130.[8] Zhang W, Yang W, Liu X, et al. Rapidly constructed scaffold-free embryonic stem cell sheets for ocular surface reconstruction. Scanning. 2014;36(3):286-292.[9] Kayama M, Kurokawa MS, Ueno H, et al. Recent advances in corneal regeneration and possible application of embryonic stem cell-derived corneal epithelial cells. Clin Ophthalmol. 2007;1(4):373-382.[10] 刘德伍,邹萍.脱细胞角膜基质与骨髓间充质干细胞复合构建组织工程化活性人工角膜[J].中国临床康复, 2006,10(25):54-56.[11] Ma Y, Xu Y, Xiao Z, et al. Reconstruction of chemically burned rat corneal surface by bone marrow-derived human mesenchymal stem cells. Stem Cells. 2006;24(2):315-321.[12] 孙红,乔丽萍,侯世科,等.骨髓间充质干细胞静注对碱烧伤兔角膜修复的促进作用及机制[J].山东医药,2017,57(20):40-42.[13] Hayashi R, Ishikawa Y, Sasamoto Y, et al. Co-ordinated ocular development from human iPS cells and recovery of corneal function. Nature. 2016;531(7594):376-380.[14] Tzameret A, Sher I, Belkin M, et al. Epiretinal transplantation of human bone marrow mesenchymal stem cells rescues retinal and vision function in a rat model of retinal degeneration. Stem Cell Res. 2015;15(2):387-394.[15] Yang C, Yang Y, Brennan L, et al. Efficient generation of lens progenitor cells and lentoid bodies from human embryonic stem cells in chemically defined conditions. FASEB J. 2010; 24(9):3274-3283.[16] Fan X, Li J, Zhu J, et al. Computer-assisted orbital volume measurement in the surgical correction of late enophthalmos caused by blowout fractures. Ophthalmic Plast Reconstr Surg. 2003;19(3):207-211.[17] Eto H, Suga H, Inoue K, et al. Adipose injury-associated factors mitigate hypoxia in ischemic tissues through activation of adipose-derived stem/progenitor/stromal cells and induction of angiogenesis. Am J Pathol. 2011;178(5): 2322-2332.[18] Lamba DA, McUsic A, Hirata RK, et al. Generation, purification and transplantation of photoreceptors derived from human induced pluripotent stem cells. PLoS One. 2010; 5(1):e8763.[19] Chou BK, Mali P, Huang X, et al. Efficient human iPS cell derivation by a non-integrating plasmid from blood cells with unique epigenetic and gene expression signatures. Cell Res. 2011;21(3):518-529.[20] Li P, Hu H, Yang S, et al. Differentiation of induced pluripotent stem cells into male germ cells in vitro through embryoid body formation and retinoic acid or testosterone induction. Biomed Res Int. 2013;2013:608728.[21] Moad M, Pal D, Hepburn AC, et al. A novel model of urinary tract differentiation, tissue regeneration, and disease: reprogramming human prostate and bladder cells into induced pluripotent stem cells. Eur Urol. 2013;64(5):753-761.[22] Warren L, Manos PD, Ahfeldt T, et al. Highly efficient reprogramming to pluripotency and directed differentiation of human cells with synthetic modified mRNA. Cell Stem Cell. 2010;7(5):618-630.[23] Shalom-Feuerstein R, Serror L, De La Forest Divonne S, et al. Pluripotent stem cell model reveals essential roles for miR-450b-5p and miR-184 in embryonic corneal lineage specification. Stem Cells. 2012;30(5):898-909.[24] Banin E, Obolensky A, Idelson M, et al. Retinal incorporation and differentiation of neural precursors derived from human embryonic stem cells. Stem Cells. 2006;24(2):246-257.[25] Haruta M, Sasai Y, Kawasaki H, et al. In vitro and in vivo characterization of pigment epithelial cells differentiated from primate embryonic stem cells.Invest Ophthalmol Vis Sci. 2004;45(3):1020-1025.[26] Lund RD, Adamson P, Sauvé Y, et al. Subretinal transplantation of genetically modified human cell lines attenuates loss of visual function in dystrophic rats. Proc Natl Acad Sci U S A. 2001;98(17):9942-9947.[27] Binder S, Krebs I, Hilgers RD, et al. Outcome of transplantation of autologous retinal pigment epithelium in age-related macular degeneration: a prospective trial. Invest Ophthalmol Vis Sci. 2004;45(11):4151-4160.[28] Lamba DA, Karl MO, Ware CB, et al. Efficient generation of retinal progenitor cells from human embryonic stem cells. Proc Natl Acad Sci U S A. 2006;103(34):12769-12774.[29] Osakada F, Ikeda H, Mandai M, et al. Toward the generation of rod and cone photoreceptors from mouse, monkey and human embryonic stem cells. Nat Biotechnol. 2008;26(2): 215-224.[30] Kicic A, Shen WY, Wilson AS, et al. Differentiation of marrow stromal cells into photoreceptors in the rat eye. J Neurosci. 2003;23(21):7742-7749.[31] Rezanejad H, Soheili ZS, Haddad F, et al. In vitro differentiation of adipose-tissue-derived mesenchymal stem cells into neural retinal cells through expression of human PAX6 (5a) gene. Cell Tissue Res. 2014;356(1):65-75.[32] 陈鹏,梁洁,钱焕文.视紫红质及RPE65蛋白在光致视网膜变性疾病中的作用机制[J].激光生物学报,2007,16(2):139-142.[33] Wernig M, Meissner A, Foreman R, et al. In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature. 2007;448(7151):318-324.[34] Meyer JS, Katz ML, Maruniak JA, et al. Embryonic stem cell-derived neural progenitors incorporate into degenerating retina and enhance survival of host photoreceptors. Stem Cells. 2006;24(2):274-283.[35] Stark MR, Biggs JJ, Schoenwolf GC, et al. Characterization of avian frizzled genes in cranial placode development. Mech Dev. 2000;93(1-2):195-200.[36] Hirano M, Yamamoto A, Yoshimura N, et al. Generation of structures formed by lens and retinal cells differentiating from embryonic stem cells. Dev Dyn. 2003;228(4):664-671.[37] Ooto S, Haruta M, Honda Y, et al. Induction of the differentiation of lentoids from primate embryonic stem cells. Invest Ophthalmol Vis Sci. 2003;44(6):2689-2693.[38] Yang C, Yang Y, Brennan L, et al. Efficient generation of lens progenitor cells and lentoid bodies from human embryonic stem cells in chemically defined conditions. FASEB J. 2010; 24(9):3274-3283.[39] Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126(4):663-676.[40] Zhou M, Leiberman J, Xu J, et al. A hierarchy of proliferative cells exists in mouse lens epithelium: implications for lens maintenance. Invest Ophthalmol Vis Sci. 2006;47(7): 2997-3003.[41] Qiu X, Yang J, Liu T, et al. Efficient generation of lens progenitor cells from cataract patient-specific induced pluripotent stem cells. PLoS One. 2012;7(3):e32612.[42] Okita K, Ichisaka T, Yamanaka S. Generation of germline-competent induced pluripotent stem cells. Nature. 2007;448(7151):313-317.[43] Jacobsson D, Panciera F, Tersoff J, et al. Interface dynamics and crystal phase switching in GaAs nanowires. Nature. 2016; 531(7594):317-322.[44] Hayashi R, Ishikawa Y, Sasamoto Y, et al. Co-ordinated ocular development from human iPS cells and recovery of corneal function. Nature. 2016;531(7594):376-380. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||