Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (8): 1281-1286.doi: 10.3969/j.issn.2095-4344.0149
Previous Articles Next Articles
DNA vaccines: mechanisms of action
Gao Xiao-pei1, Guan Xiao-yan1, 2, Bai Guo-hui 2, Liu Jian-guo1, 2
- 1School of Stomatology, Zunyi Medical University, Zunyi 563099, Guizhou Province, China; 2the Special Key Laboratory of Oral Diseases Research, Institution of Higher Education in Guizhou Province, the Key Laboratory of Oral Diseases Research of Zunyi, Zunyi 563006, Guizhou Province, China
-
Received:2017-11-23Online:2018-03-18Published:2018-03-18 -
Contact:Liu Jian-guo, M.D., Professor, School of Stomatology, Zunyi Medical University, Zunyi 563099, Guizhou Province, China; the Special Key Laboratory of Oral Diseases Research, Institution of Higher Education in Guizhou Province, the Key Laboratory of Oral Diseases Research of Zunyi, Zunyi 563006, Guizhou Province, China -
About author:Gao Xiao-pei, Master candidate, Physician, School of Stomatology, Zunyi Medical University, Zunyi 563099, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81560186; the Social Development Project of Science and Technology Department of Guizhou Province, No. SY[2013]3043; the University Key Laboratory Construction Project of Guizhou Province, No. KY[2013]109; the Joint Fund of Science and Technology Department of Guizhou Province, No. J-LKZ[2012]47
CLC Number:
Cite this article
Gao Xiao-pei1, Guan Xiao-yan1, 2, Bai Guo-hui 2, Liu Jian-guo1, 2. DNA vaccines: mechanisms of action[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(8): 1281-1286.
share this article
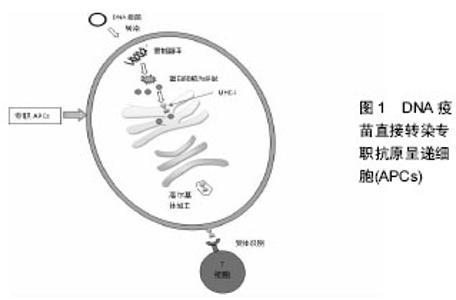
2.1 来自细菌DNA的先天免疫信号 DNA疫苗的主要优势之一是“嵌入式”的佐剂效应,即细菌DNA拥有直接激活先天免疫的能力[9]。最近的研究强调了先天免疫与适应性免疫的关联性,以及在广泛的适应性免疫应答中强大的固有信号具有重要意义[10]。哺乳动物已经进化到可以在细胞核内隔绝DNA,而且DNA的表达能大量激活多种先天免疫信号[11]。通常细菌DNA及质粒富含Toll样受体9(Toll-like receptor 9,TLR9)介导的非甲基化CpG岛。在某些情况下,非甲基化CpG基因与TLR9的结合导致Ⅰ型干扰素与促炎症细胞因子的释放。TLR9识别信号在DNA疫苗免疫原性方面可能会增强适应性免疫反应,但并不是必须的。在临床前和临床试验中研究的编码肿瘤抗原DNA疫苗,已经能利用改良的包含CpG富集序列的质粒载体来增强TLR9信号[12]。 cGAMP作为第二信使会导致细胞核区域的干扰素基因激活蛋白的二聚化和移位[13]。在Shikawa等[14]开发的抗前列腺癌DNA疫苗动物实验中,TKB和IFNar2基因敲除小鼠在疫苗接种后未能诱导产生T细胞和B细胞免疫应答,可见两种基因对DNA疫苗成功应用具有至关重要的作用。在STING基因敲除小鼠模型中研究者观察到了相似的结果,同时观察到了抗原特异性抗体的滴度降低以及IFN-γ分泌减少。然而Suchak等[15]的报告发现在体内试验中环GMP-AMP合酶(Cyclic GMP-AMP synthase,cGAS)对于DNA疫苗诱导的免疫反应是可有可无的,甚至认为cGAS可能是一种多余的STING DNA传感器上游信号。进一步的证据支持DNA疫苗诱导的免疫原性需要Ⅰ型干扰素的释放,报告还强调了适应性免疫应答中IRF7的必要性。大多数的研究报告指出在质粒DNA疫苗接种后IRF3对T细胞免疫有重要意义,而抗体免疫应答却没有[15-16]。 AIM2(absent in melanoma 2)是一种包含热蛋白结构域的干扰素诱导型DNA传感器,它最初是在黑色素瘤细胞系中发现的,它的缺失导致更加显著的细胞增殖,也增加了致癌的可能性[17]。AIM2将自体细胞质和病原体的dsDNA结合形成二聚体,使ASC利用交叉热蛋白结构域与感受器蛋白发生相互作用[18]。AIM2与ASC结合后通过CARD域的相互作用产生焦亡小体,从而在细胞中激活caspas-1诱导炎性细胞死亡。这条通路激活的caspase-1诱导了促炎细胞因子白细胞介素1β和白细胞介素18的成熟和分泌。利用Ⅰ型IFN诱导炎性细胞死亡的方式,AIM2可以通过阻断cGAS-STING和TLR9通路的信号来减少Ⅰ型IFN的产生。事实上,Liu等[19]的研究报告发现在牛分枝杆菌感染期间AIM2抑制了自噬和IFNβ的生产。Suschak等[20]评估了该途径的重要性在DNA疫苗传感方面的重要性,在白细胞介素1β和白细胞介素18依赖型的Aim2基因敲除小鼠模型中观察到了T细胞和B细胞的免疫应答受损。改变固有的DNA传感机制是一种提高DNA疫苗的免疫原性的潜在方法,要么通过使用媒介来激活不同的传感器,要么将DNA疫苗定向到单个的DNA传感器。 2.2 DNA疫苗直接转染组织细胞 DNA疫苗通过多种途径进入体内,组织细胞(如肌细胞、皮肤角朊细胞)直接摄取质粒DNA,被摄取的质粒DNA分子在特定启动子作用下,在细胞核内转录为mRNA,mRNA进而与游离核糖体相结合,再被转移至细胞质内翻译成抗原蛋白分子,表达出的抗原蛋白分子被分泌到细胞外、结合到细胞膜上或局限于细胞浆内,这些抗原蛋白经过内吞、溶酶体降解、与MHC分子结合后呈递给初始型T细胞。然后该途径诱导产生Th2淋巴细胞主导的免疫应答与获得性体液免疫,机体获得有效的保护性抗体以抵抗病原菌的侵害。例如,将DNA直接注射人肌肉可导致蛋白的表达和诱导CTL反应,研究发现培养的肌细胞通过T小管和细胞膜穴样内陷可将质粒纳入。同时黏膜免疫系统包含大量的能摄取抗原并且递呈到黏膜下淋巴组织的微皱褶细胞[21]。而在上皮下淋巴组织里,各种类型的细胞可能被转染,它们与某些共刺激分子一起参与将MHC-I与MHC-Ⅱ限制性抗原递呈给CD4+与CD8+T淋巴细 胞[22]。Smith等[23]使用表面电波仪来定位表皮细胞,通过单次剂量的皮下注射呼吸道合胞病毒(respiratorysyncytial virus,RSV)DNA疫苗即可使大鼠获得完全的抗呼吸道感染免疫力。 2.3 交叉激活和交叉呈递机制 组织细胞表达的外源蛋白凭借细胞内吞作用进入细胞质基质,作为胞内抗原被加工,由专职抗原呈递细胞(Professional antigen- presenting cell,APC)通过MHC-I类分子途径递呈给CD8+T细胞,激活抗原特异性细胞毒性T淋巴细胞(cytotoxic lymphocyte,CTL)称为交叉呈递、交叉激 活[24-25]。最早关于DNA疫苗作用机制的研究表明有一部分的细胞毒性T淋巴细胞应答是由细胞呈递激活的,反而不是那些产生的编码抗原。例如,肌注被NP基因稳定转染的肌母细胞或者编码了流感病毒核蛋白的DNA质粒均能诱导很强的细胞毒性T淋巴细胞免疫应答。使用骨髓嵌合体,同样的研究小组表明,除非NP多肽呈递给专职抗原呈递细胞的MHC分子中,否则不能产生针对NP肽的细胞毒性T淋巴细胞应答。这是第一次证明非专职抗原呈递细胞产生的抗原能够通过交叉呈递给专职抗原呈递细胞激活细胞毒性T淋巴细胞免疫应答[26]。 接着更多的研究表明与质粒中普遍存在的启动子相比,肌肉或者皮肤特异性启动子并不会降低抗原特异性免疫应答水平。而肌细胞作为体内抗原库,不断地释放抗原至细胞间隙,然后被专职抗原呈递细胞吞噬、加工、递呈,然后与这些细胞内质网内产生的MHC-Ⅱ类分子结合,然后递呈于Th2淋巴细胞,从而刺激CD8+T细胞,最终引发免疫应答[27]。Hon等[28]的研究表明使用不能在专职抗原呈递细胞表达抗原的角质细胞启动子比巨细胞病毒启动子诱导更低水平的细胞毒性T淋巴细胞应答。这些数据证明DNA疫苗接种后的抗原启动主要发生在交叉呈递途径而不是传统认为的内源性途径。Zhao等[29]构建了一种包含肿瘤表达抗原G250和细胞毒性T淋巴细胞相关的抗原4(CTLA-4)基因的DNA疫苗, 而CTLA-4是T细胞活化的一个阴性调节剂,它与CD28的结合具有更强的亲和力。使用联合表达的DNA疫苗进行接种不仅诱导产生了更高水平的抗CTLA-4和抗g250抗体,而且在小鼠中也增加了g250特异性T细胞反应。 2.4 DNA疫苗直接转染专职抗原呈递细胞 经各种途径(皮肤、黏膜、肌肉等)接种DNA疫苗,可以直接转染专职专职抗原呈递细胞,在专职抗原呈递细胞中发生转染、活化、内源性表达质粒编码的抗原。质粒DNA在专职抗原呈递细胞内表达的蛋白质抗原经胞浆内的蛋白酶降解形成8-10个氨基酸组成的肽,被内质网上的抗原转运结构(TAP)转入内质网腔,与MHC-I类分子形成聚合体,经高尔基体到达专职抗原呈递细胞膜表面,被T细胞的受体识别,这是诱导细胞毒性T淋巴细胞应答的最有效途径[30](见图1)。"

关于先天免疫的研究揭示与交叉呈递途径相比,专职抗原呈递细胞的转接转染、呈递和相关细胞内在激活会诱导更强的适应性免疫[31]。Bot等[32]已经报告了专职抗原呈递细胞能够表达质粒编码的抗原并具有体内启动能力。例如Farris、Zhang等[33-34]利用显微注射递送DNA疫苗,通过穿透皮肤表面持续释放DNA疫苗,并递送给定植在真皮内的专职抗原呈递细胞,即朗格汉斯细胞。因为朗格汉斯细胞是专职抗原呈递细胞可以被直接转染, 抗原就可以在专职抗原呈递细胞内表达并被加工、递呈, 同时产生共刺激分子[35-36]。Hu等[37]利用微针递送编码了肿瘤抗原的PEI/DNA复合体,它含有细胞穿透力的糖基化结合肽,可以经皮肤直接递送至树突状细胞,促进强烈的Th1细胞分化,同时诱导CD4+和CD8+细胞向肿瘤渗透。Wang等[38]开发了核壳氧化硅层状双氢氧化物(SiO2LDH)包裹编码了HBsAg的pDNA。在体外实验中SiO2LDH/DNA纳米复合材料有效地被巨噬细胞内吞,引起了基因的高表达。当采用非肠胃给药小鼠时,SiO2LDH/pHBsAg诱导了增强的HBV抗体和抗原特异性T细胞应答。此外,用SiO2LDH/pHBVsAg接种疫苗纳米颗粒激活巨噬细胞,并通过激活NF-苗纳途径促进Th1分化。还有研究发现在被动转移后B细胞是首选的专职专职抗原呈递细胞[39]。 在小鼠和人类系统中,将DNA被动递送给B细胞而不是树突状细胞,会导致编码抗原的呈递从而扩大同源性CD8+T细胞应答。与此同时,被动转移到树突状细胞或其他骨髓细胞的DNA在表达之前发生了明显地降 解[40]。这些发现表明将DNA疫苗靶向递送至B细胞可能是增加直接抗原表达的一种手段,或者提高DNA疫苗在树突状细胞和其他骨髓细胞中DNA溶酶体的逃逸[41]。 2.5 细胞凋亡机制 有研究发现细胞凋亡产生的凋亡小体可以有效地被树突状细胞, 朗格汉斯细胞或单核细胞捕获, 进而激发针对凋亡小体所携带抗原的免疫应答, 这种反应表现为长期的特异性的抗凋亡小体的免疫记忆。当免疫部位被转染的细胞发生凋亡,释放的凋亡小体携带抗原,使抗原高效特异性地定位于树突状细胞,通过交叉致敏呈递给CD8+ T细胞激发强烈的免疫应答[42]。 首先,Sasaki等[43]报道了衰减的caspase蛋白酶能够在流感DNA疫苗模型中诱导细胞凋亡,并产生一定范围和强度的免疫应答。一系列的研究表明通过诱导DNA疫苗转染细胞发生凋亡,能有成功提高DNA疫苗的免疫效力。当然,不是细胞凋亡水平越高,DNA疫苗的免疫应答水平越高,两者之间存在某种平衡关系。Kojima等[44]使用IRES、CMV及双启动子系统来促进caspase蛋白酶的表达,对比实验结果发现翻译水平较低的IRES启动子系统中,抗HIV-1 DNA疫苗细胞凋亡水平能够诱导产生最大程度的抗体。因此在一定细胞凋亡水平下才可以有效促进树突状细胞摄取抗原,才能够最大程度地提高DNA疫苗的免疫原性、增强免疫应答。再者,细胞凋亡常常是各种病毒感染的结果,以甲病毒复制子为基础的复制型DNA载体构建DNA疫苗,能够模拟病毒感染机体的自然过程从而诱导免疫保护[45]。例如,在RNA复制子载体系统中,启动子并不直接指导外源基因的表达,而是控制具有自主复制能力的甲病毒RNA复制子的表达,甲病毒RNA可翻译产生病毒复制酶复合物,进而介导插入的外源基因在细胞浆内大量转录复制,使得外源基因编码的mRNA和蛋白获得高效表达。RNA复制子的大量复制可导致被转染细胞的凋亡,凋亡的细胞可被树突状细胞等其他抗原递呈细胞摄取、吸收,选择递呈免疫系统。同时, 外源基因在表达过程中会产生双链RNA中间体,其能产生很多的免疫刺激信号,并对树突状细胞的直接效应诱导其成熟活化,提高MHC分子的表达。Wang、Zheng等[46-47]均使用RNA复制子作为DNA疫苗载体在小鼠模型中进行了相关试验,研究发现在血清中特异性IgG、细胞因子IFN-γ、白细胞介素2、白细胞介素10的表达显著高于对照组,同时诱导产生了大量的CD8+T细胞,获得了有效的细胞免疫和体液免疫应答。除此之外,因被转染细胞会直接凋亡能够有效提升DNA疫苗的安全性。 DNA疫苗的作用机制见表1。"

| [1] MA L.DNA vaccines: an historical perspective and view to the future.Immunol Reviews.2011,293(1):62-84.[2] Li L,Saade F,Petrovsky N.The future of human DNA vaccines.J Biotechnol.2012;162(2-3):171-182.[3] Ren Y, Wang N, Hu W, et al.Successive site translocating inoculation potentiates DNA/recombinant vaccinia vaccination.Sci Rep.2015;5:18099.[4] Zahm CD,Colluru VT, McNeel DG.DNA vaccines for prostate cancer. Pharmacol Ther.2017.174:27-42.[5] Hannaman D, Dupuy LC, Ellefsen B,et al.A Phase 1 clinical trial of a DNA vaccine for Venezuelan equine encephalitis delivered by intramuscular or intradermal electroporation.Vaccine. 2016;34(31): 3607-3612.[6] Li J,Mo Z,Li G,et al.Generation and evaluation of virulence attenuated mutants of Edwardsiella tarda as vaccine candidates to combat edwardsiellosis in flounder (Paralichthys olivaceus). Fish Shellfish Immunol.2015;43(1):175-180.[7] Christopher D.Zahm, V. T., Douglas G.McNeel.Technologies for enhanced efficacy of DNA vaccines.Expert Reviews Vaccines. 2012;11:189-209.[8] Barber GN.Innate immune DNA sensing pathways: STING, AIMII and the regulation of interferon production and inflammatory responses.Curr Opin Immunol.2011;23(1):10-20.[9] Desmet CJ, Ishii KJ.Nucleic acid sensing at the interface between innate and adaptive immunity in vaccination.Nat Rev Immunol. 2012;12(7):479-491.[10] Kobiyama K, Jounai N,Aoshi T,et al.Innate Immune Signaling by, and Genetic Adjuvants for DNA Vaccination.Vaccines (Basel). 2013;1(3):278-292.[11] Nie Y,Wang YY.Innate immune responses to DNA viruses.Protein Cell.2013;4(1):1-7.[12] Chen SP, Peng RH, Chiou PP.Modulatory effect of CpG oligodeoxynucleotide on a DNA vaccine against nervous necrosis virus in orange-spotted grouper (Epinephelus coioides).Fish Shellfish Immunol.2015;45(2):919-926.[13] Rekoske BT,Smith HA, Olson BM,et al.PD-1 or PD-L1 Blockade Restores Antitumor Efficacy Following SSX2 Epitope-Modified DNA Vaccine Immunization.Cancer Immunol Res.2015;3(8): 946-955.[14] Ishikawa H,Ma Z,Barber GN.STING regulates intracellular DNA-mediated, type I interferon-dependent innate immunity. Nature.2009;461(7265):788-792.[15] Suschak JJ,Wang S,Fitzgerald KA,et al.A cGAS-Independent STING/IRF7 Pathway Mediates the Immunogenicity of DNA Vaccines.J Immunol.2016;196(1):310-316.[16] Shirota H,Petrenko L,Hattori T,et al.Contribution of IRF-3 mediated IFNbeta production to DNA vaccine dependent cellular immune responses.Vaccine.2009;27(15):2144-2149.[17] Ratsimandresy RA, Dorfleutner A,Stehlik C.An Update on PYRIN Domain-Containing Pattern Recognition Receptors: From Immunity to Pathology.Front Immunol.2013;4:440.[18] Fernandes-Alnemri T,Yu JW, Datta P, et al.AIM2 activates the inflammasome and cell death in response to cytoplasmic DNA.Nature.2009;458(7237):509-513.[19] Liu C,Yue R,Yang Y,et al.AIM2 inhibits autophagy and IFN-β production during M. bovis infection.Oncotarget.2016;7(30): 46972-46987.[20] Suschak JJ, Wang S, Fitzgerald KA,et al.Identification of Aim2 as a sensor for DNA vaccines.J Immunol.2015;194(2):630-636.[21] 秦妍,管晓燕,白国辉,等.基因疫苗黏膜免疫及其机制的研究进展[J].贵州医药,2016,40(4):426-428.[22] Walters JN, Ferraro B, Duperret EK,et al.A Novel DNA Vaccine Platform Enhances Neo-antigen-like T Cell Responses against WT1 to Break Tolerance and Induce Anti-tumor Immunity.Mol Ther.2017;25(4):976-988.[23] Smith TR, Schultheis FK, Morrow MP,et al.Development of an intradermal DNA vaccine delivery strategy to achieve single-dose immunity against respiratory syncytial virus.Vaccine.2017;35(21): 2840-2847.[24] Johnson LE, Frye TP,McNeel DG.Immunization with a prostate cancer xenoantigen elicits a xenoantigen epitope-specific T-cell response.Oncoimmunology. 2012;1(9):1546-1556.[25] Lambert L,Kinnear E, McDonald JU,et al.DNA Vaccines Encoding Antigen Targeted to MHC Class II Induce Influenza-Specific CD8(+) T Cell Responses, Enabling Faster Resolution of Influenza Disease.Front Immunol.2016;7:321.[26] UlmerJ, Otten G.Priming of CTL responses by DNA vaccines-direct transfection of antigen presenting cells versus cross-priming.Developmental Biology.2000;104:9-14.[27] 李敏,董竞男,刘建国,等.基因疫苗作用机制和免疫途径的研究现状[J].贵州医药,2013,37(8):748-751.[28] Hon H,Oran A, Brocker T, et al.B Lymphocytes Participate in Cross-Presentation of Antigen following Gene Gun Vaccination.The Journal of Immunology.2005;174(9):5233-5242.[29] Zhao Y,Wei Z,Yang H,et al.Enhance the anti-renca carcinoma effect of a DNA vaccine targeting G250 gene by co-expression with cytotoxic T-lymphocyte associated antigen-4(CTLA-4). Biomedicine & Pharmacotherapy. 2017;90:147-152.[30] Ashok M, Rangarajan P.Immunization with DNA Plasmids Coding for Crimean-Congo Hemorrhagic Fever Virus Capsid and Envelope Proteins andor Virus-Like Particles Induces Protection and Survival in Challenged Mice.Vaccine.2002;20(12):1563-1570.[31] Iwasaki A,Medzhitov R.Regulation of adaptive immunity by the innate immune system.Science.2010;327(5963):291-295.[32] Bot A,Stan AC,Inaba K,et al.Dendritic cells at a DNA vaccination site express the encoded influenza nucleoprotein and prime MHC class I-restricted cytolytic lymphocytes upon adoptive transfer. International Immunology.2000;12(6):825-832.[33] Farris E,Brown DM,Ramer-Tait AE,et al.Micro- and nanoparticulates for DNA vaccine delivery.Exp Biol Med (Maywood). 2016;241(9):919-929.[34] Zhang M,Hong Y,Chen W,et al.Polymers for DNA Vaccine Delivery.ACS Biomaterials Sci Eng.2016;3(2):108-125.[35] Fernando GJ, Zhang J, Ng HI,et al.Influenza nucleoprotein DNA vaccination by a skin targeted, dry coated, densely packed microprojection array (Nanopatch) induces potent antibody and CD8(+) T cell responses.J Control Release.2016;237:35-41.[36] Watkins C,Hopkins J,Harkiss G.Reporter gene expression in dendritic cells after gene gun administration of plasmid DNA. Vaccine.2005;23(33):4247-4256.[37] Hu Y,Xu B,Xu J,et al.Microneedle-assisted dendritic cell-targeted nanoparticles for transcutaneous DNA immunization.Polym Chem. 2015;6(3):373-379.[38] Wang J,Zhu R,Gao B,et al.The enhanced immune response of hepatitis B virus DNA vaccine using SiO2@LDH nanoparticles as an adjuvant.Biomaterials.2014;35(1):466-478.[39] Gerloni M,Rizzi M,Castiglioni P,et al.T cell immunity using transgenic B lymphocytes. Proc Natl Acad Sci U S A. 2004; 101(11):3892-3897.[40] Colluru VT, McNee DG.B lymphocytes as direct antigen-presenting cells for anti-tumor DNA vaccines.Oncotarget. 2016;7(42):67901-67918.[41] Garu A, Moku G, Gulla SK, et al.Genetic Immunization With In Vivo Dendritic Cell-targeting Liposomal DNA Vaccine Carrier Induces Long-lasting Antitumor Immune Response.Mol Ther.2016; 24(2):385-397.[42] 刘浩.凋亡基因与DNA疫苗[J].现代免疫学,2004,24(6):520-522.[43] SasakiS, Xin KQ, Okudela K,et al.Immunomodulation by apoptosis-inducing caspases for an influenza DNA vaccine delivered by gene gun.Gene Therapy.2002;9(12):828-831.[44] Kojima Y, Jounai N, Takeshita F,et al.The degree of apoptosis as an immunostimulant for a DNA vaccine against HIV-1 infection. Vaccine.2007;25(3):438-445.[45] 李平忠.DNA疫苗和细胞凋亡[J].国外医学:预防诊断治疗用生物制品分册,2004,27(5):218-220.[46] Wang Y, Yue X, Jin H, et al.A suicidal DNA vaccine expressing the fusion protein of peste des petits ruminants virus induces both humoral and cell-mediated immune responses in mice.J Virol Methods.2015;225:35-40.[47] Zheng L, Hu Y, Hua Q, et al.Protective immune response in mice induced by a suicidal DNA vaccine encoding NTPase-II gene of Toxoplasma gondii.Acta Trop.2017;166:336-342. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [3] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [4] | Liu Bo, Chen Xianghe, Yang Kang, Yu Huilin, Lu Pengcheng. Mechanism of DNA methylation in exercise intervention for osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 791-797. |
| [5] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [6] | Zhao Xiang, Wei Cuilan, Zhang Yeting. Neurogenesis and neuroinflammation under exercise: alteration and regulation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 813-820. |
| [7] | Chen Junyi, Wang Ning, Peng Chengfei, Zhu Lunjing, Duan Jiangtao, Wang Ye, Bei Chaoyong. Decalcified bone matrix and lentivirus-mediated silencing of P75 neurotrophin receptor transfected bone marrow mesenchymal stem cells to construct tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 510-515. |
| [8] | Ma Zhijie, Li Jingyu, Cao Fang, Liu Rong, Zhao Dewei. Influencing factors and biological property of novel biomedical materials: porous silicon carbide coated with bioactive tantalum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 558-563. |
| [9] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [10] | Shi Xiaoxiu, Mao Shilong, Liu Yang, Ma Xingshuang, Luo Yanfeng. Comparison of tantalum and titanium (alloy) as orthopedic materials: physical and chemical indexes, antibacterial and osteogenic ability [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 593-599. |
| [11] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [12] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [13] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [14] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [15] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||