Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (22): 3594-3601.doi: 10.12307/2023.374
Previous Articles Next Articles
Influence of hip-preservation surgeries on subsequent total hip arthroplasty for osteonecrosis of the femoral head
Wang Zhangzheng1, Mo Liang1, Zhou Chi1, Liu Yuhao1, Yan Hongsong1, He Wei1, 2, 3
- 1First Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510405, Guangdong Province, China; 2Hip Research Center of Guangzhou University of Chinese Medicine, Guangzhou 510405, Guangdong Province, China; 3Guangdong Academy of Traditional Chinese Medicine Orthopedics, Guangzhou 510378, Guangdong Province, China
-
Received:2022-02-07Accepted:2022-07-07Online:2023-08-08Published:2022-11-03 -
Contact:He Wei, MD, Professor, First Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510405, Guangdong Province, China; Hip Research Center of Guangzhou University of Chinese Medicine, Guangzhou 510405, Guangdong Province, China; Guangdong Academy of Traditional Chinese Medicine Orthopedics, Guangzhou 510378, Guangdong Province, China -
About author:Wang Zhangzheng, Master candidate, First Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510405, Guangdong Province, China -
Supported by:General Project of National Natural Science Foundation of China, No. 81873327 (to HW); Scientific Research Project of Guangdong Provincial Administration of Traditional Chinese Medicine, No. 20221136 (to LYH); Innovation and Strong Hospital Project of First Affiliated Hospital of Guangzhou University of Chinese Medicine, No. 2019IIT06 (to HW)
CLC Number:
Cite this article
Wang Zhangzheng, Mo Liang, Zhou Chi, Liu Yuhao, Yan Hongsong, He Wei. Influence of hip-preservation surgeries on subsequent total hip arthroplasty for osteonecrosis of the femoral head[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(22): 3594-3601.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
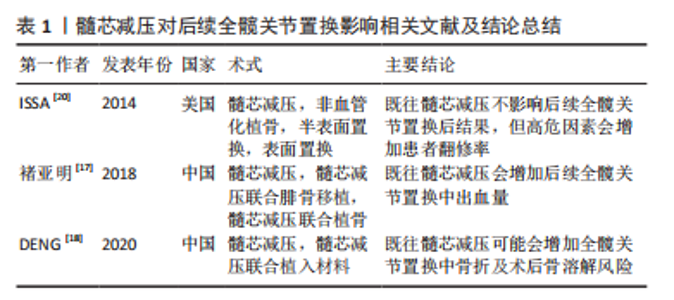
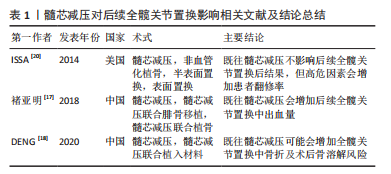
2.1 各种保髋手术对后续全髋关节置换的影响 2.1.1 髓芯减压 髓芯减压最早由Ficat和Arlet于1962年提出。该手术通过钻孔减压来降低股骨头内部的局部压力,为新生血管提供空间,同时刺激周围微血管的形成,增强坏死区骨组织的爬行替代[9]。 据研究报道,单纯髓芯减压保髋治疗的10年生存率约为50%[12-13]。因此目前单纯的髓芯减压已较少使用,为了提高疗效,一些学者尝试在髓芯减压的同时植入钽棒、自体骨或同种异体骨或骨髓间充质干细胞等生物材料。 而关于既往单纯髓芯减压对后续全髋关节置换影响的相关文献较少,部分研究将髓芯减压和髓芯减压联合植入材料纳入同一组的分组方式有待商榷[17-18]。例如褚亚明等[17]的研究指出既往髓芯减压会增加后续全髋关节置换中出血量,但此结论可能受到该研究中将单纯髓芯减压和髓芯减压联合腓骨移植纳入同一组的分组方式的影响。 髋关节功能评分和假体存活率反映了全髋关节置换后的临床结果和行翻修手术的风险[19],是研究者首要关注的指标,褚亚明等[17]和ISSA等[20]的研究表明既往髓芯减压手术史不会影响后续全髋关节置换后假体存活率与术后Harris髋关节评分,最近的一项荟萃分析也得出了相同的结论[18]。同时,ISSA等[20]的研究中既往髓芯减压手术史的患者和无既往保髋手术史接受全髋关节置换的患者在影像学评估和并发症等方面均无显著性差异。髓芯减压的手术方式是利用环钻或克氏针在股骨颈进行钻孔减压[8],虽然不可避免的会造成股骨头及转子间区域骨小梁分布发生改变[17],这可能会增加全髋关节置换中转子间骨折的风险,但由于其较小的侵入性且较少的影响股骨近端结构,对后续全髋关节置换影响较小,但仍应注意术中骨折、术后骨溶解的风险[17-18,20],见表1。"
2.1.2 非血管化植骨 单纯髓芯减压由于缺乏对软骨下板的结构支撑,无法提供合适的力学修复环境,因此有学者提出髓芯减压联合植骨治疗早期股骨头坏死。髓芯减压减压钻孔形成的骨隧道也给髓芯减压联合植骨的产生创造了条件;同时为了更好地清除头内死骨,也产生了诸如“活板门技术”和“电灯泡技术”等的术式[21]。 在钻孔减压基础上植入非血管化骨材料(钽棒、陶瓷骨棒,自体或同种异体腓骨、自体或同种异体骨碎屑或骨髓间充质干细胞)可以为坏死区的修复提供力学支撑与生物基础。此类术式在随后全髋关节置换中存在截取股骨头后残留植入材料影响开髓、扩髓及股骨柄假体放置的问题而增加手术的难度。蔡剑等[22]的研究中就指出既往髓芯减压联合腓骨移植患者行全髋关节置换时,需要用咬骨钳与箱式截骨刀清理皮质骨残留,部分患者可能还需要使用高速磨钻进行打磨,该研究发现既往髓芯减压联合腓骨移植患者有更长的平均手术时间和切口平均长度。褚亚明等[17]的研究则发现髓芯减压联合植骨会增加后续全髋关节置换中的出血量。这些研究结论可以从某些方面说明髓芯减压联合腓骨移植增加了后续全髋关节置换手术难度。但现有的研究均表明髓芯减压联合腓骨移植失败的患者与初次治疗即选择全髋关节置换的患者在术后Harris评分结果、髋臼杯外展角及股骨假体中置率方面无差异[17,22]。综合目前研究,髓芯减压联合不带血管的骨移植可能会一定程度上增加后续全髋关节置换的难度,但不会影响全髋关节置换后的髋关节功能恢复,在影像学评估中也不会造成不良影响。 相较于髓芯减压联合的非血管化植骨,“活板门技术”和“电灯泡技术”等技术虽然能够更好地清除头内死骨,但是需要更大的手术视野,从而会造成更大的手术创伤[21],但已有的研究认为打压植骨并不会对后续全髋关节置换造成不利影响。ZUO等[23]的一项回顾性研究表明打压植骨失败后全髋关节置换的患者和初次治疗即选择全髋关节置换的患者在术后Harris评分和影像学评估方面无差异。RIJNEN等[24]的研究也表明打压植骨失败术后接受全髋关节置换患者仍能得到较好的临床结果,末次随访时(平均随访4年)平均Harris评分为91分,仅有6%(1/17)的并发症发生率(1髋出现了无菌性松动并翻修)。 综合上述研究结果可见,非血管化植骨后续的全髋关节置换能够有效恢复髋关节功能,并且其不会影响全髋关节置换后影像学评估结果。虽然有文献报道非血管化植骨会增加后续全髋关节置换的难度,但全髋关节置换仍可以视作非血管化植骨保髋失败后的可靠治疗手段。 2.1.3 带血运的植骨 常规的非血管化的骨移植手术清理死骨之后,会在坏死区植骨填充以提供力学支撑。但是移植骨并不带有血供且植入位置血供较差,不利于血管长入,易导致植入物的吸收与囊变。为改善这种情况,临床上出现了大量不同方式的带血运植骨,通过向股骨头内提供血供,为新骨的生成创造有利条件。目前临床上主要包括带蒂髂骨瓣及带血管腓骨移植[25]。 吻合血管游离腓骨移植对后续全髋关节置换患者的预后影响存在争议,BEREND等[26]评价了73例(85髋)吻合血管游离腓骨移植失败后进行全髋关节置换患者的预后,并进行了9年的随访,该研究认为吻合血管游离腓骨移植后全髋关节置换功能改善效果与之前报道的无既往保髋手术史并接受全髋关节置换的年轻患者效果相当。但DAVIS等[27]报道在第3年随访时,与之前未进行吻合血管游离腓骨移植的全髋关节置换后患者相比,吻合血管游离腓骨移植组髋关节功能评分有显著性差异(P=0.03)。 吻合血管游离腓骨移植同样存在移植物残留影响股骨柄假体放置的问题,移植物的取出方式会影响到全髋关节置换的效果。DAVIS等[27]发现未进行广泛的移植物切除时,全髋关节置换后股骨柄内翻畸形的发生率很高;但当使用高速磨钻去除残余移植物时,虽然股骨柄不齐率显著下降,但全髋关节置换中出血量(P=0.017)与手术时间(P=0.000 2)显著增加。 既往的吻合血管游离腓骨移植还会造成更高的并发症发生率,RYAN等[28]表明吻合血管游离腓骨移植不仅会增加后续全髋关节置换手术时间和出血量(P < 0.001),同时增加血肿形成(P=0.004)、术中骨折(P=0.053)以及深部感染(P=0.044)的发生率。 既往带血运髂骨移植不会对后续全髋关节置换后假体存活率造成影响。CHAU等[29]的研究发现带血运髂骨移植+全髋关节置换组(27例,37髋)和单纯全髋关节置换组(242例,302髋)的总体假体存活率无显著性差异(P=0.71)。 值得一提的是,CHAU等[29]的研究虽然认为带血运髂骨移植不会对后续全髋关节置换后假体存活率造成影响,但该研究同时指出吸烟或饮酒的全髋关节置换患者有更低的假体存活率和更短的假体存活时间:吸烟者或饮酒者的平均假体存活时间为23.14年;非吸烟者或饮酒者为30.48年(P < 0.01)。BEREND等[26]的研究发现酒精诱导的股骨头坏死患者有更高的假体翻修率(P < 0.05)。ISSA等[20]研究也发现具有饮酒、吸烟、系统性红斑狼疮、HIV、镰状细胞病等危险因素的高风险组患者在保髋手术失败后接受全髋关节置换会有更高概率需要进行翻修手术。目前的大部分研究报道纳入的样本量较少,且少有研究考虑到这些因素,这可能会影响到这些研究的结论。 带血运的植骨后续全髋关节置换可能更具挑战性。手术时间延长及失血增加,可能与移植物的切除和髋关节周围软组织的损伤有关。尽管带血运的植骨失败患者的全髋关节置换后功能预后评分可能相对较低且有更高的并发症发生率,但考虑到全髋关节置换能够有效提升带血运的植骨失败患者的髋关节功能,且术后的假体存活率不会受到既往带血运的植骨的影响,患者仍然可以从全髋关节置换中获得很大的好处。 作者将植骨对后续全髋关节置换影响的相关研究作一归纳,见表2。"
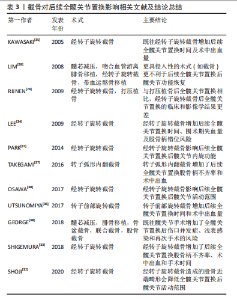
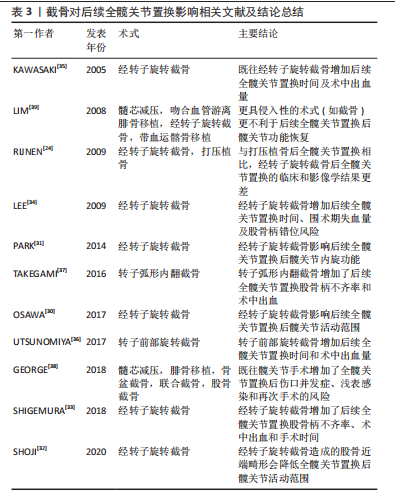
2.1.4 截骨 截骨最早是由日本Sugioka医生首先介绍的经转子旋转截骨,其核心思想是通过截骨的方式,将股骨头负重坏死区或塌陷区转移至非负重区,使正常骨结构作为新负重区起到力学支撑作用,从而延缓甚至避免关节面塌陷,并改善头臼不匹配状态[9]。保髋截骨主要包括经转子弧形内翻截骨和经转子旋转截骨等。 既往研究显示经转子旋转截骨会造成后续全髋关节置换后更小的髋关节活动范围,OSAWA等[30]进行了一项术后随访平均10年的临床研究,发现经转子旋转截骨组的髋关节屈曲、外展和内旋功能明显低于原始全髋关节置换组。PARK等[31]的研究也发现既往经转子旋转截骨影响后续全髋关节置换后髋关节内旋功能。SHOJI等[32]利用三维建模的计算方法说明了经转子旋转截骨造成的股骨近端畸形会降低全髋关节置换后髋关节活动范围。但在Harris评分结果方面仍存在部分争议,SHIGEMURA等[31,33]的研究认为既往经转子旋转截骨不会影响后续全髋关节置换后Harris评分得分,但在OSAWA等[30]研究中经转子旋转截骨组[(83.3±8.7)分]和初次全髋关节置换组[(89.3±7.6)分]的Harris评分在末次随访时差异显著(P < 0.01),但该研究中经转子旋转截骨失败后接受全髋关节置换患者的Harris评分结果仍有80分以上的平均分,并且两组的生活质量评分无显著差异。基于上述文献,文章认为尽管研究结果存在争议,并且会造成髋关节活动范围的减小,全髋关节置换仍可较好地改善经转子旋转截骨失败后患者的髋关节功能。 经转子旋转截骨会导致股骨近端结构改变,造成后续全髋关节置换手术的困难。有研究报道了经转子旋转截骨后续的全髋关节置换手术有更长的手术时间和更多的出血量[30,33-35]。同时这种结构的改变也可能对股骨柄假体的位置造成不良影响,研究表明既往经转子旋转截骨手术史的患者较初次全髋关节置换患者术后有更高股骨柄假体不齐率[30,33-34]。而股骨柄假体的移位很自然会进一步导致患者下肢长度的差异增大[30]。 尽管既往经转子旋转截骨会造成股骨柄假体不齐率的增加,但这种改变并不会影响到假体的稳定性。SHIGEMURA等[33]的一项荟萃分析表明,既往经转子旋转截骨手术史不会增加后续全髋关节置换后的脱位率。有研究报道了既往经转子旋转截骨手术史的患者同初次全髋关节置换患者在种植体稳定性方面无差异[34-35]。KAWASAKI等[35]同样报道了经转子旋转截骨不会影响随后全髋关节置换后的假体存活率。 转子前部旋转截骨和转子弧形内翻截骨的相关文献结果同经转子旋转截骨类似。UTSUNOMIYA等[36]的研究表明既往转子前部旋转截骨手术史不影响后续全髋关节置换后Harris评分,但截骨后组的患者随后的全髋关节置换手术时间更长,术中失血量更大,并且患肢加长的情况更明显。TAKEGAMI等[37]的研究则发现转子弧形内翻截骨后全髋关节置换和初次全髋关节置换在髋关节功能评分方面无差异,但转子弧形内翻截骨组患者全髋关节置换手术出血量增加且有更高的股骨柄不齐率。 在假体脱位、感染和翻修等并发症发生率方面,一些研究认为既往的截骨手术史不会造成后续全髋关节置换更高的并发症发生率[30,33-34,37]。但该结论仍然存在争议,GEORGE等[38]将34例(37髋)既往有髋关节手术史并接受全髋关节置换的患者(其中22髋为既往截骨患者)与无既往髋关节手术史并接受的全髋关节置换患者进行比较,发现既往的保髋手术不会影响30岁以下年轻患者后续全髋关节置换后假体的5年存活率,但是会造成他们有更高的伤口并发症(P=0.037)、浅表感染(P=0.005)和再次手术(P=0.015)的发生率。 鲜有文献报道既往保髋手术会造成后续全髋关节置换后更差的假体存活率和髋关节功能评分。但既往更具有侵入性的保髋手术的确会造成更大的创伤以及更广泛的组织粘连,进而影响随后全髋关节置换的短期疗效。LIM等[39]对30例(36髋)保髋手术失败的患者进行了平均55个月的随访,其中包含23髋接受了微创保髋手术(髓芯减压)与13髋经历更具有创伤性保髋手术(经转子旋转截骨2髋、吻合血管游离腓骨移植6髋,带血运髂骨移植 5髋),并将他们与直接进行全髋关节置换的患者(39髋)相比较,结果发现前者有更长的全髋关节置换手术时间,更多的术中出血量及总出血量,以及更小的髋关节活动范围,特别是在更具创伤性的保髋手术组表现更为明显,但二者在Harris评分、假体存活率上无显著差异,见表3。 "

截骨改变股骨近端的结构并在术中造成广泛的组织粘连从而增加后续全髋关节置换手术操作的难度,这表现在已有诸多的研究报道截骨后的全髋关节置换会有更长的手术时间与更多的出血量。股骨近端的结构同样可能影响到术者操作过程中对假体放置位置、角度的评估,以及影响到术后假体的稳定。全髋关节置换植入假体持久的固定需要假体部件和支撑骨之间实现紧密的几何形状匹配[40]。髋臼杯错位会导致脱位风险增加、活动范围受限和撞击征[40],Lewinnek安全区[髋臼倾斜(40±10)°,前倾(15±10)°]常作为髋臼假体放置位置的评估标准[41],BEVERLAND 等[42]发现Lewinnek安全区内髋臼假体的脱位率为1.5%,安全区外髋臼假体的脱位率为6.1%。因此部分研究会将髋臼杯前倾角、髋臼杯外展角作为全髋关节置换后影像学评估的组成部分。而股骨柄的稳定性取决于从植入假体到股骨近端及远端力量负荷转移的平衡[40]。冠状面股骨柄错位可能会影响患肢腿长与臀肌功能进而不利于后续全髋关节置换的临床效果[43-44]。多个研究报道了截骨后续全髋关节置换后有更高的股骨柄不齐率、更大的腿长差异以及更小的短期髋关节活动范围。但对基于Harris评分的全髋关节置换后髋功能和假体存活率方面,截骨似乎并不会产生不利影响。 2.1.5 钽棒植入 多孔钽棒具有蜂窝状结构且弹性模量与正常骨骼接近,该材料具有良好的生物相容性用于清除头内死骨后置入以作为支撑物。多孔钽植入也是现今用于保髋治疗的术式之一。 钽棒的取出增加了多孔钽植入后续全髋关节置换手术的难度,并且可能会增加股骨术中骨折风险。ZUO等[23]将多孔钽植入后全髋关节置换的患者同初次全髋关节置换的患者和打压植骨术后全髋关节置换的患者相比较,发现多孔钽植入后全髋关节置换的手术时间和术中出血量较后两组明显更高,并且认为该结构和钽棒取出的过程相关。这与LEE等[45-46]研究结论一致。钽棒的取出方式有多种,常规方式是通过摆锯在股骨颈处截骨并切断钽棒,而后使用环锯通过顺行或逆行的方向取出剩余部分[45]。也有选择克氏针在钽棒周围钻孔,钽棒松动后被完整的取出[23]。多孔钽棒具有良好的生物相容性,这导致了钽棒取出的技巧要求很高从而增加了全髋关节置换的难度[46-49]。无论选择哪一种取出方式都不可避免地导致沿钽棒插入轨迹特别是股骨外侧皮质的骨流失,进而导致股骨骨折风险的增加和出血量的增多并且延长了手术时间。股骨近端骨质流失可能会导致骨折和假体不稳定,但尚无有力证据证明多孔钽植入失败患者全髋关节置换后有更高的术中骨折率和股骨柄下沉发生率[23,46]。作者总结了钽棒植入对后续全髋关节置换的相关进度,见表4。"


即使在C臂的引导下,取出钽棒时从一端到另一端维持稳定的速率与保持和植入钽棒相同的轨迹都很困难,并且常规的钽棒取出方法甚至需要通过摆锯截断钽棒,因此钽碎片的生成是不可避免的。多孔钽植入失败后行全髋关节置换后钽碎片的影响也引起了研究者们的关注。OLSEN等[47]研究了21例(21髋)多孔钽植入后接受全髋关节置换患者术后的X射线片,发现大多数钽碎片分布在股骨区;但与年龄、性别与之相匹配的21例(21髋)初次全髋关节置换的患者相比,二者在假体线性磨损率方面并无差异,各时间段随访的Harris评分和线性磨损率之间并无相关性。CHENG等[48]的研究在钽碎片主要分布区域和对假体线性磨损率的影响方面得出了相同的结论,但他认为残留的钽碎片增加了骨溶解的风险,甚至可能引发炎症反应破坏成骨过程,这表现在该研究中多孔钽植入组股骨假体周围骨溶解和放射通透性显著性的高于对照组。短期随访的研究表明钽碎片的残留不会对随后全髋关节置换预后造成不良影响,但其对关节磨碎速率的长期影响和造成局部炎症反应引发骨溶解进而导致假体松动甚至脱位的风险,仍需要更多的长期随访研究提供证据。 现有的研究认为多孔钽植入对随后的全髋关节置换后功能恢复和假体生存率方面无影响[45-48],最近一项平均8年随访的回顾性研究报道了多孔钽植入联合髓芯减压和同种异体骨移植治疗早期或有限病变范围的股骨头坏死患者有较好的生存率(96个月总生存率为75%)[49],且转归组全髋关节置换后5年Harris评分(平均89分)与原发性全髋关节置换组Harris评分(平均92分)之间无显著差异(P=0.09)。而对如大腿疼痛、感染、假体脱位及假体周围骨折等,目前的研究同样不认为既往多孔钽植入手术史会增加上述并发症的发生率[45-46,48]。此外现有研究认为既往多孔钽植入史对于全髋关节置换后髋臼假体外展角、髋臼假体前倾角并无影响[45-46,48],见表4。 综合目前的研究,多孔钽植入不会影响后续全髋关节置换后的髋关节功能及假体生存率。相较未有保髋手术史的全髋关节置换患者,有多孔钽植入手术史的全髋关节置换患者的影像学评估结果和并发症发生率并无差异。但多孔钽植入对后续全髋关节置换的影响目前仍存在争议。一方面,钽棒的去除会增加全髋关节置换手术的难度,导致失血量的增加、手术时间的延长,且容易导致骨质的流失进而增加股骨骨折的风险,特别是转子骨折风险的增高可能会导致假体不稳定。另一方面,任何移除方法都不可避免的产生金属碎屑,虽然目前未有证据证明残留的金属碎屑会造成不利影响,但其仍存在加速假体磨损和导致骨质溶解的风险。 2.2 髋关节表面置换对后续全髋关节置换的影响 髋关节表面置换是全髋关节置换的一种替代方案。相较于传统的全髋关节置换,髋关节表面置换保留了股骨骨量和股骨近端结构,从而使重建后的髋关节应力分布同人体生理状态更相符合,同时更大尺寸的股骨头关节使重建后的髋关节更为稳定,这两个优势使得髋关节表面置换特别适合髋关节活动活跃的年轻患者[50]。髋关节表面置换采用仿生学制作人体髋臼帽及股骨头杯覆盖等关节面,理论上应该被视为髋关节重建的一种手术方式而非髋关节保留手术,但考虑到髋关节表面置换作为髋关节手术的一种,并且因为感染、股骨颈骨折、局部软组织反应等原因,髋关节表面置换可能需要行转换为全髋关节置换的翻修手术。故在此也将髋关节表面置换对于后续全髋关节置换的影响纳入讨论。 既往髋关节表面置换并不会对后续全髋关节置换后的临床结果已经假体存活率造成不良影响,ISSA等[20]的一项平均75个月的随访研究表明,表面置换组和半表面置换组患者同无既往髋关节手术的对照组在Harris评分和全髋关节置换后假体存活率方面无差异(P > 0.05)。ESWARAMOORTHY等[51-53]的研究同样也报道了相同的结论。而MCGRATH等[53]的研究则发现髋关节表面置换会增加后续全髋关节置换手术时间。髋关节表面置换转换为全髋关节置换的临床结果与翻修原因有关,SU等[54]的研究表明因机械原因(如股骨颈骨折、假体松动)进行翻修的患者有最高的全髋关节置换 术后Harris评分结果(平均为99分),并且显著高于Harris评分结果最低的不明原因疼痛/金属敏感组(平均为86.9分,P=0.029)。上述研究具体内容见表5。 "

| [1] ZHAO D, ZHANG F, WANG B, et al. Guidelines for clinical diagnosis and treatment of osteonecrosis of the femoral head in adults (2019 version). J Orthop Transl. 2020;21:100-110. [2] LEARMONTH ID, YOUNG C, RORABECK C. The operation of the century:total hip replacement. Lancet. 2007;370(9597):1508-1519. [3] ZHAO DW, YU M, HU K, et al. Prevalence of nontraumatic osteonecrosis of the femoral head and its associated risk factors in the chinese population: results from a nationally representative survey. Chin Med J (Engl). 2015; 128(21):2843-2850. [4] TAN B, LI W, ZENG P, et al. Epidemiological study based on china osteonecrosis of the femoral head database. Orthop Surg. 2021;13(1): 153-160. [5] 赵德伟,谢辉. 成人股骨头坏死保髋手术治疗的策略及探讨[J].中国修复重建外科杂志,2018,32(7):792-797. [6] 郭玉玲.中国人均预期寿命时空变化及影响因素分析[J].中国卫生政策研究,2018,11(8):44-49. [7] HUANG ZQ, FU FY, LI WL, et al. Current treatment modalities for osteonecrosis of femoral head in mainland China: a cross-sectional study. Orthop Surg. 2020;12(6):1776-1783. [8] 朱诗白,张啸天,陈曦,等.股骨头坏死的保髋治疗[J].中华关节外科杂志(电子版),2020,14(6):741-746. [9] 刘予豪,周驰,陈雷雷,等.基于股骨头坏死围塌陷期理论的保髋术式总结[J].中国修复重建外科杂志,2017,31(8):1010-1015. [10] HERNIGOU P, DUBORY A, HOMMA Y, et al. Cell therapy versus simultaneous contralateral decompression in symptomatic corticosteroid osteonecrosis:a thirty year follow-up prospective randomized study of one hundred and twenty five adult patients. Int Orthop. 2018;42(7):1639-1649. [11] OSAWA Y, SEKI T, OKURA T, et al. Long-term outcomes of curved intertrochanteric varus osteotomy combined with bone impaction grafting for non-traumatic osteonecrosis of the femoral head. Bone Joint J. 2021; 103-B(4):665-671. [12] MARTINOT P, DARTUS J, LECLERC JT, et al. Hip survival after plain core decompression alone versus bone morphogenetic protein and/or bone marrow reinjection with core decompression for avascular osteonecrosis of the femoral head: a retrospective case control study in ninety two patients. Int Orthop. 2020;44(11):2275-2282. [13] LI M, MA Y, FU G, et al. 10-year follow-up results of the prospective, double-blinded, randomized, controlled study on autologous bone marrow buffy coat grafting combined with core decompression in patients with avascular necrosis of the femoral head. Stem Cell Res Ther. 2020;11(1),287. [14] XIE H, WANG B, TIAN S, et al. Retrospective long-term follow-up survival analysis of the management of osteonecrosis of the femoral head with pedicled vascularized iliac bone graft transfer. J Arthroplasty. 2019;34(8): 1585-1592. [15] XU Y, REN Y, ZHAO Z, et al. Hip survival rate in the patients with avascular necrosis of femoral head after transtrochanteric rotational osteotomy: a systematic review and meta-analysis. Chin Med J. 2019;132(24):2960-2971. [16] AURÉGAN J, VILLAIN B, BÉGUÉ T. What is the rate of patients undergoing a total hip arthroplasty after core decompression and insertion of a tantalum rod in osteonecrosis of the femoral head: a systematic review. Int Orthop. 2018;42(7):1631-1638. [17] 褚亚明,张亮,张金庆,等.全髋关节置换治疗保髋失败的非创伤性股骨头缺血性坏死的中期随访结果[J].中华骨与关节外科杂志,2018, 11(8):596-599. [18] DENG W, WANG Z, ZHOU Y, et al. Does prior core decompression have detrimental effect on subsequent total hip arthroplasty for osteonecrosis of the femoral head: a systematic review and meta-analysis. Orthop Traumatol Surg Res. 2020;106(8):1553-1560. [19] SINGH JA, SCHLECK C, HARMSEN S, et al. Clinically important improvement thresholds for Harris Hip Score and its ability to predict revision risk after primary total hip arthroplasty. BMC Musculoskelet Disord. 2016;17:256. [20] ISSA K, JOHNSON AJ, NAZIRI Q, et al. Hip osteonecrosis: does prior hip surgery alter outcomes compared to an initial primary total hip arthroplasty? J Arthroplasty. 2014;29(1):162-166. [21] 陈铭璁,傅煊健,陈扬.股骨头减压非血管化植骨保头手术进展[J].中国骨与关节损伤杂志,2020,35(10):1115-1116. [22] 蔡剑,岳永彬,孙海忠,等.股骨头坏死异体腓骨移植失败后的全髋关节置换[J].东南大学学报(医学版),2018,37(4):588-593. [23] ZUO W, MA JH, CUI W, et al. Comparison of total hip arthroplasty after two types of failed hip preserving procedures with primary total hip arthroplasty. Orthop Surg. 2020;12(1):162-169. [24] RIJNEN WHC, LAMEIJN N, SCHREURS BW, et al. Total hip arthroplasty after failed treatment for osteonecrosis of the femoral head. Orthop Clin North Am. 2009;40(2):291-298. [25] 丁海波,王阳,陆芸.带血管骨移植治疗股骨头坏死的研究进展[J].中国修复重建外科杂志,2021,35(3):381-386. [26] BEREND KR, GUNNESON E, URBANIAK JR, et al. Hip arthroplasty after failed free vascularized fibular grafting for osteonecrosis in young patients. J Arthroplasty. 2003;18(4):411-419. [27] DAVIS ET, MCKEE MD, WADDELL JP, et al. Total hip arthroplasty following failure of free vascularized fibular graft. J Bone Joint Surg Am. 2006;88 Suppl 3:110-115. [28] RYAN SP, WOOSTER B, JIRANEK W, et al. Outcomes of conversion total hip arthroplasty from free vascularized fibular grafting. J Arthroplasty. 2019; 34(1):88-92. [29] CHAU W, NG JP, LAU H, et al. Osteonecrosis of the hip: is there a difference in the survivorship of total hip arthroplasty with or without previous vascular iliac bone grafting? J Orthop Surg Res. 2021;16(1):244. [30] OSAWA Y, SEKI T, MORITA D, et al. Total hip arthroplasty after transtrochanteric rotational osteotomy for osteonecrosis of the femoral head: a mean 10-year follow-up. J Arthroplasty. 2017;32(10):3088-3092. [31] PARK KS, TUMIN M, PENI I, et al. Conversion total hip arthroplasty after previous transtrochanteric rotational osteotomy for osteonecrosis of the femoral head. J Arthroplasty. 2014;29(4):813-816. [32] SHOJI T, YAMASAKI T, OTA Y, et al. Effect of transtrochanteric rotational osteotomy on impingement and contact state of a femoral implant in conversion total hip arthroplasty-Retrospective simulation study. Clin Biomech (Bristol, Avon). 2020;71:68-72. [33] SHIGEMURA T, YAMAMOTO Y, MURATA Y, et al. Total hip arthroplasty after failed transtrochanteric rotational osteotomy for osteonecrosis of the femoral head: a systematic review and meta-analysis. Orthop Traumatol Surg Res. 2018;104(8):1163-1170. [34] LEE YK, HA YC, KIM KC, et al. Total hip arthroplasty after previous transtrochanteric anterior rotational osteotomy for femoral head osteonecrosis. J Arthroplasty. 2009;24(8):1205-1209. [35] KAWASAKI M, HASEGAWA Y, SAKANO S, et al. Total hip arthroplasty after failed transtrochanteric rotational osteotomy for avascular necrosis of the femoral head. J Arthroplasty. 2005;20(5):574-579. [36] UTSUNOMIYA T, MOTOMURA G, IKEMURA S, et al. The results of total hip arthroplasty after sugioka transtrochanteric anterior rotational osteotomy for osteonecrosis. J Arthroplasty. 2017;32(9):2768-2773. [37] TAKEGAMI Y, KOMATSU D, SEKI T, et al. Total hip arthroplasty after failed curved intertrochanteric varus osteotomy for avascular necrosis of the femoral head. Nagoya J Med Sci. 2016;78(1):89-97. [38] GEORGE J, MILLER EM, HIGUERA CA, et al. Influence of prior hip salvage surgery on outcomes after total hip arthroplasty in young patients. J Arthroplasty. 2018;33(4):1108-1112. [39] LIM S, MOON Y, EUN S, et al. Total hip arthroplasty using the s-rom modular stem after joint-preserving procedures for osteonecrosis of the femoral head. J Arthroplasty. 2008;23(4):495-501. [40] YANG Z, JIAN W, LI ZH, et al. The geometry of the bone structure associated with total hip arthroplasty. PLoS One. 2014;9(3):e91058. [41] 朱东明,张振,张杰,等.全髋关节置换后假体脱位危险因素的最新进展[J]中国组织工程研究,2020,24(36):5864-5870. [42] BEVERLAND DE, O’NEILL CKJ, RUTHERFORD M, et al. Placement of the acetabular component. Bone Joint J. 2016;98BA(1):37-43. [43] FOTTNER A, WOICZINSKI M, KISTLER M, et al. Varus malalignment of cementless hip stems provides sufficient primary stability but highly increases distal strain distribution. Clin Biomech (Bristol, Avon). 2018;58:14-20. [44] HAVERSATH M, BUSCH A, JAGER M, et al. The ‘critical trochanter angle’: a predictor for stem alignment in total hip arthroplasty. J Orthop Surg Res. 2019;14(1):165. [45] MA J, WANG B, YUE D, et al. Outcomes of conversion tha after failed porous tantalum implant for osteonecrosis of the femoral head:a comparative matched study. HIP International. 2020;30(6):703-710. [46] LEE G, PARK K, KIM D, et al. Results of total hip arthroplasty after core decompression with tantalum rod for osteonecrosis of the femoral head. Clin Orthop Surg. 2016;8(1):38-44. [47] OLSEN M, LEWIS PM, MORRISON Z, et al. Total hip arthroplasty following failure of core decompression and tantalum rod implantation. Bone Joint J. 2016;98-B(9):1175-1179. [48] CHENG Q, TANG J, GU J, et al. Total hip arthroplasty following failure of tantalum rod implantation for osteonecrosis of the femoral head with 5- to 10-year follow-up. BMC Musculoskel Disord. 2018;19(1):289. [49] HE M, WEI Q, CHEN Z, et al. Porous tantalum rod implantation is associated with low survival rates in patients with type C2 osteonecrosis of the femoral head but has no effect on the clinical outcome of conversion total hip arthroplasty: a retrospective study with an average 8-year follow-up. BMC Musculoskel Disord. 2020;21(1):841. [50] MARSH M, NEWMAN S. Trends and developments in hip and knee arthroplasty technology. J Rehabil Assist Technol Eng. 2021;8:2055668320952043. [51] ESWARAMOORTHY VK, BIANT LC, FIELD RE. Clinical and radiological outcome of stemmed hip replacement after revision from metal-on-metal resurfacing. J Bone Joint Surg Br. 2009;91(11):1454-1458. [52] BALL ST, LE DUFF MJ, AMSTUTZ HC. Early results of conversion of a failed femoral component in hip resurfacing arthroplasty. J Bone Joint Surg Am. 2007;89(4):735-741. [53] MCGRATH MS, MARKER DR, SEYLER M, et al. Surface replacement is comparable to primary total hip arthroplasty. Clin Orthop Relat Res. 2009; 467(1):94-100. [54] SU EP, SU SL. Surface replacement conversion: results depend upon reason for revision. Bone Joint J. 2013;95-B(11 Suppl A):88-91. [55] 顾长源,杜斌,孙光权,等.股骨头坏死保髋治疗常用术式的利与弊. 中国组织工程研究,2020,24(21):3379-3386. [56] FEHRLE MJ, CALLAGHAN JJ, CLARK CR, et al. Uncemented total hip arthroplasty in patients with aseptic necrosis of the femoral head and previous bone grafting. J Arthroplasty. 1993;8(1):1-6. [57] BOACHIE-ADJEI Y, KO JK, CUI Q. Total hip arthroplasty after failed operative treatment for osteonecrosis of the femoral head. Seminars in Arthroplasty. 2008;19(4):267-273. [58] PIERCE TP, ELMALLAH RK, JAUREGUI JJ, et al. Outcomes of total hip arthroplasty in patients with osteonecrosis of the femoral head-a current review. Curr Rev Musculoskelet Med. 2015;8(3):246-251. |
| [1] | Wu Taoguang, Nie Shaobo, Chen Hua, Zhu Zhengguo, Qi Lin, Tang Peifu. Biomechanical characteristics of a new multi-dimensional cross locking plate in the treatment of subtrochanteric nonunion [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1330-1334. |
| [2] | Ke Yuqi, Chen Changjian, Wu Hao, Zheng Lianjie. Comparison of 12-month follow-up results of primary total hip arthroplasty between modified direct anterior approach and direct anterior approach [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1377-1382. |
| [3] | He Yinhao, Li Xiaosheng, Chen Hongwen, Chen Tiezhu. 3D printed porous tantalum metal in the treatment of developmental dysplasia of the hip: current status and application prospect [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1455-1461. |
| [4] | Yang Jiujie, Li Zhi, Wang Shujie, Tian Ye, Zhao Wei. Intraoperative neurophysiological monitoring of functional changes following durotomy with decompression for acute spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1232-1236. |
| [5] | Zhao Wei, Feng Wei, Yang Tieyi, Ren Wei, Wang Yuxin, Lyu Huicheng, Chang Zhiqiang, Feng Xiaodong, Wang Ziheng, Guo Shibing. Antibiotic bone cement intramedullary nail prepared using 3D printed mold for the treatment of long bone infection in lower limbs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1023-1030. |
| [6] | Li Hui, Zhang Kun, Hao Yangquan, Feng Lei, Yang Zhi, Xu Peng, Lu Chao. Robot-assisted core decompression and bone grafting for ARCO II osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 547-551. |
| [7] | Yu He, Zheng Jiafa, Song Xiufeng, Guan Shengyi. Tibiotalocalcaneal arthrodesis with blood supplied fibular flap combined with hollow screw in the treatment of end-stage ankle osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 588-593. |
| [8] | Zhang Hui, Wang Jiayang, Wang Qian, Gan Hongquan, Wang Zhiqiang. Effects of hyaluronic acid combined with domestic porous tantalum on chondrocyte function under the dynamic environment [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 339-345. |
| [9] | Liu Huan, Li Han, Ma Yunhao, Zhong Weijian, Ma Guowu. Osteogenic capacity of partially demineralized dentin particles in the maxillary sinus lift [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 354-359. |
| [10] | You Aijia, Li Wenjie, Zhou Junli, Li Chun. Systematic evaluation of six dressings on wound safety following total hip and knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 486-492. |
| [11] | Li Jie, Cao Shuai, Guo Dong, Zhang Qiongchi, He Xijing, Li Haopeng, Lu Teng. Finite element analysis of polyetheretherketone and titanium rods in posterior lumbar interbody fusion [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(22): 3445-3450. |
| [12] | Zhou Hongxing, Zhang Baojian, Yuan Xiangsheng. Recent evaluation of individual biological acetabular cup application in hip revision [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(22): 3521-3525. |
| [13] | Cha Xiaofeng, He Zhiyu, Wang Bo, Wu Donghai, Chen Wei, Liu Yilai, Chen Xiaojun. Model bone experiment of distal locking placement with femoral intramedullary nail [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(22): 3526-3530. |
| [14] | Luo Di, Liang Xuezhen, Yan Bozhao, Li Jiacheng, Xu Bo, Li Gang. Mechanism of Bushen Huoxue Capsule in repair of bone defects due to steroid-induced osteonecrosis of the femoral head in rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 184-191. |
| [15] | Wei Tengfei, He Xiaoming, Wei Yurou, Zhan Zhiwei, He Mincong, He Wei, Wei Qiushi. Differential expression of Piezo1 in osseous tissue of steroid- and alcohol-induced osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 270-275. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

