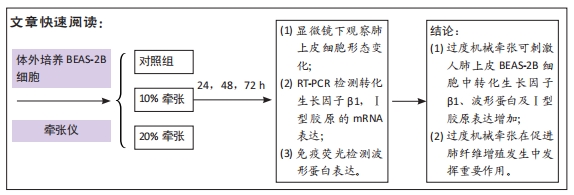
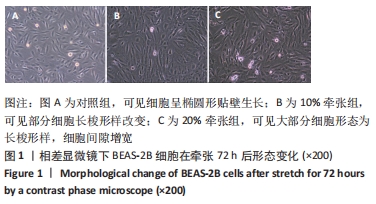
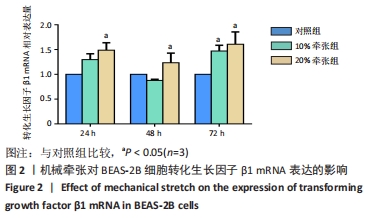
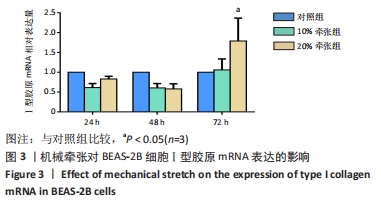
[1] MARINI JJ, ROCCO PRM, GATTINONI L. Static and Dynamic Contributors to Ventilator-induced Lung Injury in Clinical Practice. Pressure, Energy, and Power. Am J Respir Crit Care Med. 2020;201(7):767-774.
[2] GAVER DP 3RD, NIEMAN GF, GATTO LA, et al. The POOR Get POORer: A Hypothesis for the Pathogenesis of Ventilator-induced Lung Injury. Am J Respir Crit Care Med. 2020;202(8):1081-1087.
[3] BURNHAM EL, JANSSEN WJ, RICHES DW, et al. The fibroproliferative response in acute respiratory distress syndrome: mechanisms and clinical significance. Eur Respir J. 2014;43(1):276-285.
[4] NEWTON CA, HERZOG EL. Molecular Markers and the Promise of Precision Medicine for Interstitial Lung Disease. Clin Chest Med. 2021; 42(2):357-364.
[5] JAIN M. Fibroproliferative ARDS in the Era of low-tidal-volume ventilation. Chest. 2014;146(5):1140-1142.
[6] KIM KK, SHEPPARD D, CHAPMAN HA. TGF-β1 Signaling and Tissue Fibrosis. Cold Spring Harb Perspect Biol. 2018;10(4):a022293.
[7] FROESE AR, SHIMBORI C, BELLAYE PS, et al. Stretch-induced Activation of Transforming Growth Factor-beta1 in Pulmonary Fibrosis. Am J Respir Crit Care Med. 2016;194(1):84-96.
[8] LODYGA M, HINZ B. TGF-β1 - A truly transforming growth factor in fibrosis and immunity. Semin Cell Dev Biol. 2020;101:123-139.
[9] BAI L, LI A, GONG C, et al. Protective effect of rutin against bleomycin induced lung fibrosis: Involvement of TGF-β1/α-SMA/Col I and III pathway. Biofactors. 2020;46(4):637-644.
[10] ROCCO PRM, MARINI JJ. What have we learned from animal models of ventilator-induced lung injury? Intensive Care Med. 2020;46(12):2377-2380.
[11] SLUTSKY AS. History of Mechanical Ventilation. From Vesalius to Ventilator-induced Lung Injury. Am J Respir Crit Care Med. 2015; 191(10):1106-1115.
[12] JAMAATI H, NAZARI M, DAROOEI R, et al. Role of shear stress in ventilator-induced lung injury. Lancet Respir Med. 2016;4(8):e41-e42.
[13] PAPAZIAN L, DODDOLI C, CHETAILLE B, et al. A contributive result of open-lung biopsy improves survival in acute respiratory distress syndrome patients. Crit Care Med. 2007;35(3):755-762.
[14] MARTIN C, PAPAZIAN L, PAYAN MJ, et al. Pulmonary fibrosis correlates with outcome in adult respiratory distress syndrome. A study in mechanically ventilated patients. Chest. 1995;107(1):196-200.
[15] TSCHUMPERLIN DJ, OSWARI J, MARGULIES AS. Deformation-induced injury of alveolar epithelial cells. Effect of frequency, duration, and amplitude. Am J Respir Crit Care Med. 2000;162(2 Pt 1):357-362.
[16] ZHANG R, PAN Y, FANELLI V, et al. Mechanical Stress and the Induction of Lung Fibrosis via the Midkine Signaling Pathway. Am J Respir Crit Care Med. 2015;192(3):315-323.
[17] DING Z, YUAN J, LIANG Y, et al. Ryanodine Receptor Type 2 Plays a Role in the Development of Cardiac Fibrosis under Mechanical Stretch Through TGFβ-1. Int Heart J. 2017;58(6):957-961.
[18] HAMZEH MT, SRIDHARA R, ALEXANDER LD. Cyclic stretch-induced TGF-β1 and fibronectin expression is mediated by β1-integrin through c-Src- and STAT3-dependent pathways in renal epithelial cells. Am J Physiol Renal Physiol. 2015;308(5):F425-436.
[19] CHAPMAN HA, WEI Y, MONTAS G, et al. Reversal of TGFβ1-Driven Profibrotic State in Patients with Pulmonary Fibrosis. N Engl J Med. 2020;382(11):1068-1070.
[20] ZHANG Y, DAI Y, RAMAN A, et al. Overexpression of TGF-β1 induces renal fibrosis and accelerates the decline in kidney function in polycystic kidney disease. Am J Physiol Renal Physiol. 2020;319(6):F1135-F1148.
[21] REN H, ZUO S, HOU Y, et al. Inhibition of α1-adrenoceptor reduces TGF-β1-induced epithelial-to-mesenchymal transition and attenuates UUO-induced renal fibrosis in mice. FASEB J. 2020;34(11):14892-14904.
[22] ANDUGULAPATI SB, GOURISHETTI K, TIRUNAVALLI SK, et al. Biochanin-A ameliorates pulmonary fibrosis by suppressing the TGF-β mediated EMT, myofibroblasts differentiation and collagen deposition in in vitro and in vivo systems. Phytomedicine. 2020;78:153298.
[23] GONZÁLEZ-LÓPEZ A, ALBAICETA GM. Repair after acute lung injury: molecular mechanisms and therapeutic opportunities. Crit Care. 2012; 16(2):209.
[24] GONZÁLEZ-LÓPEZ A, ASTUDILLO A, GARCÍA-PRIETO E, et al. Inflammation and matrix remodeling during repair of ventilator-induced lung injury. Am J Physiol Lung Cell Mol Physiol. 2011;301(4):L500-509.
[25] CHAUHAN C, ROTH KA. Understanding Lung Development, Injury, and Repair. Am J Pathol. 2016;186(10):2518.
[26] WALLACH-DAYAN SB, ROJAS M. Senescence, the Janus of Lung Injury and Repair. Am J Respir Cell Mol Biol. 2020;62(5):548-549.
[27] ZHAO Y, RIDGE K, ZHAO J. Acute Lung Injury, Repair, and Remodeling: Pulmonary Endothelial and Epithelial Biology. Mediators Inflamm. 2017;2017:9081521.
[28] CHANDA D, OTOUPALOVA E, SMITH SR, et al. Developmental pathways in the pathogenesis of lung fibrosis. Mol Aspects Med. 2019;65:56-69.
[29] PELOSI P, ROCCO PR. Effects of mechanical ventilation on the extracellular matrix. Intensive Care Med. 2008;34(4):631-639.
[30] BREW N, HOOPER SB, ALLISON BJ, et al. Injury and repair in the very immature lung following brief mechanical ventilation. Am J Physiol Lung Cell Mol Physiol. 2011;301(6):L917-926.
[31] BITTERMAN PB. Pathogenesis of fibrosis in acute lung injury. Am J Med. 1992;92(6A):39S-43S.
[32] STRIETER RM, MEHRAD B. New mechanisms of pulmonary fibrosis. Chest. 2009;136(5):1364-1370.
[33] WILLIS BC, DUBOIS RM, BOROK Z. Epithelial origin of myofibroblasts during fibrosis in the lung. Proc Am Thorac Soc. 2006;3(4):377-382.
[34] PARAMBIL JG, MYERS JL, AUBRY MC, et al. Causes and prognosis of diffuse alveolar damage diagnosed on surgical lung biopsy. Chest. 2007; 132(1):50-57.
|