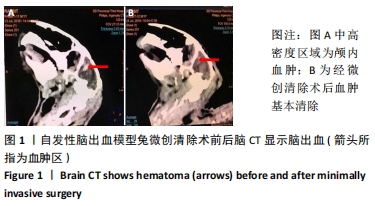
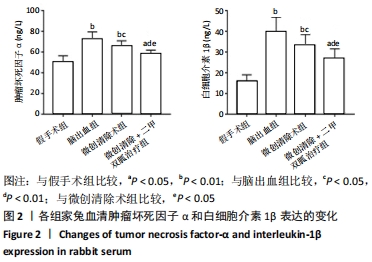
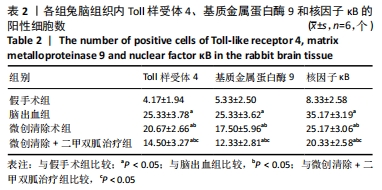
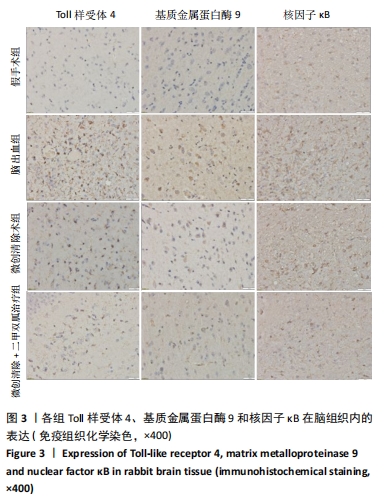
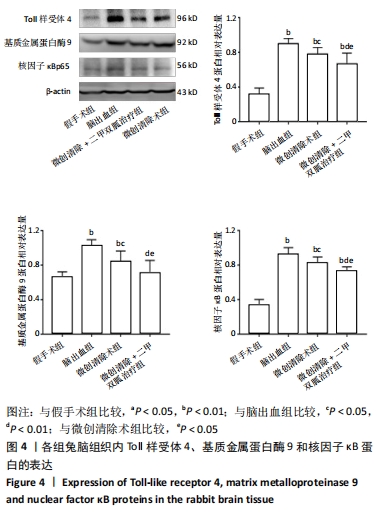
[1] ISABEL CH, DAVID JS, DAVID JW. Intracerebral hemorrhage: an update ondiagnosis and treatment. Expert Rev Neurother. 2019;19:679-694.
[2] SCAGGIANTE J, ZHANG X, MOCCO J, et al. Minimally Invasive Surgery forIntracerebral Hemorrhage. Stroke. 2018;49(11):2612-2620.
[3] HANLEY DF, THOMPSON RE, MUSCHELLI J, et al. Safety and efficacy of minimally invasive surgery plus alteplase in intracerebral haemorrhage evacuation (MISTIE): a randomised, controlled, open-label, phase 2 trial. Lancet Neurol. 2016;15(12):1228-1237.
[4] TANG Y, YIN F, FU D, et al. Efficacy and safety of minimal invasive surgery treatment in hypertensive intracerebral hemorrhage: a systematic review and meta-analysis. BMC Neurol. 2018;18:136.
[5] DUCRUET AF, HICKMAN ZL, ZACHARIA BE, et al. Exacerbation of perihematomal edema and sterile meningitis with intraventricular administration of tissue plasminogen activator in patients with intracerebral hemorrhage. Neurosurgery. 2010;66(5):648-655.
[6] LIAN LF, XU F, TANG ZP, et al. Intraclot recombinant tissuetype plasminogen activator reduces perihematomal edema and mortality in patients with spontaneous intracerebral hemorrhage. J Huazhong Univ Sci Technolog Med Sci. 2014;34(2):165-171.
[7] FATEMI I, SAEED-ASKARI P, HAKIMIZADEH E, et al. Long-term metformin therapy improves neurobehavioral functions and antioxidative activity after cerebral ischemia/reperfusion injury in rats. Brain Res Bull. 2020; 163:65-71.
[8] TAO L, LI D, LIU H, et al. Neuroprotective effects of metformin on traumatic brain injury in rats associated with NF-kappaB and MAPK signaling pathway. Brain Res Bull. 2018;140:154-161.
[9] QI B, HU L, ZHU L, et al. Metformin Attenuates Neurological Deficit after Intracerebral Hemorrhage by Inhibiting Apoptosis, Oxidative Stress and Neuroinflammation in Rats. Neurochem Res. 2017;42(10):2912-2920.
[10] 徐林,齐宏顺,李培培,等. 微创清除术联合二甲双胍对家兔脑出血病灶周围TNF-α、IL-1β和Iba-1的影响[J].神经损伤与功能重建, 2020,15(8):435-438.
[11] SAWYER CH, EVERETT JW, GREEN JD. The rabbit diencephalons in stereotaxic coordinate. J Comp Neyrol. 1954;102:801-824.
[12] PURDY PD, DEVOUS MD SR, BATJER HH, et al. Microfibrillar collagen model of canine cerebral infarction. Stroke. 1989;20(10):1361-1367.
[13] 王娜. 微创穿刺血肿引流术联合尿激酶治疗基底节区脑出血的效果[J]. 临床医学研究与实践,2020,5(29):67-68.
[14] 李少泉, 彭文龙, 张维, 等. 微创颅内血肿清除术联合术后尿激酶溶解治疗基底核区高血压脑出血的效果及对神经功能、预后的影响研究[J].解放军医药杂志 ,2020,32(11):89-93.
[15] 李飞, 高伟伟, 徐新, 等. 二甲双胍对脑出血模型小鼠血肿周围脑组织炎症反应的影响[J]. 天津医药,2017,45(5):485-488.
[16] KARIMIPOUR M, SHOJAEIZARGHANI S, MOHAJERMILANI M, et al. Pre-Treatment with Metformin in Comparison with Post-Treatment Reduces Cerebral Ischemia Reperfusion Induced Injuries in Rats. Bull Emerg Trauma. 2018;6(2):115-121.
[17] HOU X, SONG J, LI XN, et al. Metformin reduces intracellular reactive oxygen species levels by upregulating expression of the antioxidant thioredoxin via the AMPK-FOXO3 pathway. Biochem Biophys Res Commun. 2010;396(2):199-205.
[18] SAISHO Y. Metformin and inflammation: its potential beyond glucose-lowering effect. Endocr Metab Immune Disord Drug Targets. 2015; 15(3):196-205.
[19] ZHU XC, JIANG T, ZHANG QQ, et al. Chronic Metformin Preconditioning Provides Neuroprotection via Suppression of NF-kappa B-Mediated Inflammatory Pathway in Rats with Permanent Cerebral Ischemia. Mol Neurobiol. 2015;52(1):375-385.
[20] ZHOU Q, LIN FF, LIU SM, et al. Influence of the total saponin fraction from Dioscorea nipponica Makino on TLR2/4-IL1R receptor singnal pathway in rats of gouty arthritis. J Ethnopharmacol. 2017;206:274-282.
[21] JOOSTEN LA, ABDOLLAHI-ROODSAZ S, DINARELLO CA, et al. Toll like receptors and chronic inflammation in rheumatic diseases: new developments. Nat Rev Rheumatol. 2016;12(6):344-357.
[22] CRISAN TO, CLEOPHAS MC, OOSTING M, et al. Soluble uric acid primes TLR-induced proinflammatory cytokine production by human primary cells via inhibition of IL-1Ra. Ann Rheum Dis. 2016;75(4):755-762.
[23] FATTORI V, ZARPELON AC, STAURENGO-FERRARI L, et al. Budlein A, a sesquiterpene lactone from viguiera robusta, alleviates pain and inflammation in a model of acute gout arthritis in mice. Front Pharmacol. 2018;9:1076.
[24] GAO W, ZHAO Z, YU G, et al. VEGI attenuates the inflammatory injury and disruption of blood-brain barrier partly by suppressing the TLR4/NF-κB signaling pathway in experimental traumatic brain injury. Brain Res. 2015;1622(24):230 -239.
[25] CUI L, FENG L, ZHANG ZH, et al. The anti-inflammation effect of baicalin on experimental colitis through inhibiting TLR4/NF-κB pathway activation. Int Immunopharmacol. 2014;23(1):294-303.
[26] KIM C, CHO SK, KIM KD, et al. β-Caryophyllene oxide potentiates TNFα-induced apoptosis and inhibits invasion through down-modulation of NF-κB-regulated gene products. Apoptosis. 2014;19(4):708-718.
[27] JIANG X, ANDJELKOVIC AV, ZHU L, et al. Blood-brain barrier dysfunction and recovery after ischemic stroke. Prog Neurobiol. 2018;163-164: 144-171.
[28] LU Y, ZHANG XS, ZHANG ZH, et al. Peroxiredoxin 2 activates microglia by interacting with Toll-like receptor 4 after subarachnoid hemorrhage.J Neuroinflammation. 2018;15(1):87.
[29] 茅磊, 王汉东. 基质金属蛋白酶MMP-2、MMP-9在中枢神经系统中的作用[J]. 医学研究生学报,2009,22(9):992-995.
[30] OKADA T, LEI L, NISHIKAWA H, et al. TAK-242, Toll-Like Receptor 4 Antagonist, Attenuates Brain Edema in Subarachnoid Hemorrhage Mice. Acta Neurochir Suppl. 2020;127:77-81.
[31] OKADA T, SUZUKI H. Toll-like receptor 4 as a possible therapeutic target for delayed brain injuries after aneurismal subarachnoid hemorrhage. Neural Regen Res. 2017;12(2):193-196.
[32] EGASHIRA Y, ZHAO H, HUA Y, et al. White Matter Injury After Subarachnoid Hemorrhage: Role of Blood-Brain Barrier Disruption and Matrix Metalloproteinase-9. Stroke. 2015;46(10):2909-2915.
[33] OKADA T, KAWAKITA F, NISHIKAWA H, et al. Selective Toll-like receptor 4 antagonists prevent acute blood-brain barrier disruption after subarachnoid hemorrhage in mice. Mol Neurobiol. 2019;56(2):976-985.
|