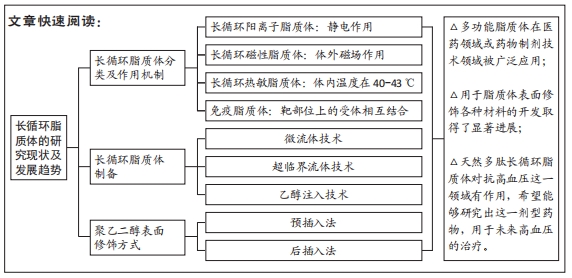
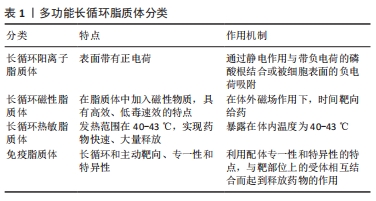
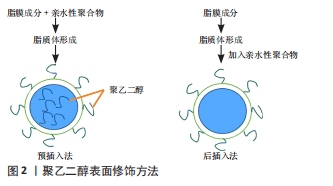
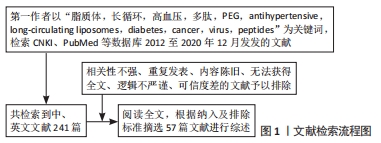
[1] LI M, DU C, GUO N, et al. Composition design and medical application of liposomes. Eur J Med Chem. 2019;164:640-653.
[2] SHAH S, DHAWAN V, HOLM R, et al. Liposomes: Advancements and innovation in the manufacturing process. Adv Drug Deliv Rev. 2020;154-155:102-122.
[3] KALYANE D, RAVAL N, MAHESHWARI R, et al. Employment of enhanced permeability and retention effect (EPR): Nanoparticle-based precision tools for targeting of therapeutic and diagnostic agent in cancer. Mater Sci Eng C Mater Biol Appl. 2019;98:1252-1276.
[4] ZHEN S, LI X. Liposomal delivery of CRISPR/Cas9. Cancer Gene Ther. 2020; 27(7-8):515-527.
[5] YE H, ZHOU L, JIN H, et al. Sorafenib-Loaded Long-Circulating Nanoliposomes for Liver Cancer Therapy. Biomed Res Int. 2020;2020:1351046.
[6] WANG X, WANG Q, LIU Z, et al. Preparation, pharmacokinetics and tumour-suppressive activity of berberine liposomes. J Pharm Pharmacol. 2017;69(6):625-632.
[7] DU Y, WANG A, WANG T, et al. Improved anti-tumor activity of novel redox-responsive paclitaxel-encapsulated liposomes based on disulfide phosphatidylcholine. Mol Pharma. 2019;17(1)262-273.
[8] Sarzani R, Spannella F, Giulietti F, et al. Cardiac Natriuretic Peptides, Hypertension and Cardiovascular Risk. High Blood Press Cardiovasc Prev. 2017;24(2):115-126.
[9] 郭晶莹,高雪萍,李永斌,等.抗高血压药物的研究新进展[J].山东化工,2015,44(22):65-67.
[10] JAIN A, JAIN A, GULBAKE A, et al. Peptide and protein delivery using new drug delivery systems. Crit Rev Ther Drug Carrier Syst. 2013;30(4):293-329.
[11] ABACHI S, BAZINET L, BEAULIEU L. Antihypertensive and Angiotensin-I-Converting Enzyme (ACE)-Inhibitory Peptides from Fish as Potential Cardioprotective Compounds. Mar Drugs. 2019;17(11):613.
[12] LEE SY, HUR SJ. Antihypertensive peptides from animal products, marine organisms, and plants. Food Chem. 2017;228:506-517.
[13] YU F, ZHANG Z, LUO L, et al. Identification and Molecular Docking Study of a Novel Angiotensin-I Converting Enzyme Inhibitory Peptide Derived from Enzymatic Hydrolysates of Cyclina sinensis. Mar Drugs. 2018;16(11):411.
[14] 艾鑫宇,袁雷.靶向流感病毒相关蛋白的小分子抑制剂研究进展[J].中国药物化学杂志,2020,30(9):549-563.
[15] 李曼,李海伟,梁烁斌,等.靶向流感病毒进入(细胞)抑制剂研究进展[J].中国科学:化学,2018,48(11):1347-1364.
[16] 蔡岩,丁晓丽,章聚宝,等.抗流感病毒药物合成研究纵览[J].化学教育(中英文),2019,40(12):5-18.
[17] TAHARA K, KOBAYASHI M, YOSHIDA S, et al. Effects of cationic liposomes with stearylamine against virus infection. Int J Pharm. 2018;543(1-2):311-317.
[18] YARIBEYGI H, ASHRAFIZADEH M, HENNEY NC, et al. Neuromodulatory effects of anti-diabetes medications: A mechanistic review. Pharmacol Res. 2020;152:104611.
[19] PETERSMANN A, MÜLLER-WIELAND D, MÜLLER UA, et al. Definition, Classification and Diagnosis of Diabetes Mellitus. Exp Clin Endocrinol Diabetes. 2019;127(S 01):S1-S7.
[20] LI W, HUANG E, GAO S. Type 1 Diabetes Mellitus and Cognitive Impairments: A Systematic Review. J Alzheimers Dis. 2017;57(1):29-36.
[21] RODRIGUEZ-FERNANDEZ S, PUJOL-AUTONELL I, BRIANSO F, et al. Phosphatidylserine-Liposomes Promote Tolerogenic Features on Dendritic Cells in Human Type 1 Diabetes by Apoptotic Mimicry. Front Immunol. 2018;9:253. doi:10.3389/fimmu.2018.00253.
[22] LANDGRAF R, ABERLE J, BIRKENFELD AL, et al. Therapy of Type 2 Diabetes. Exp Clin Endocrinol Diabetes. 2019;127(S 01):S73-S92.
[23] LEITNER DR, FRÜHBECK G, YUMUK V, et al. Obesity and Type 2 Diabetes: Two Diseases with a Need for Combined Treatment Strategies - EASO Can Lead the Way. Obes Facts. 2017;10(5):483-492.
[24] 季芳.糖尿病药物治疗的研究进展[J].山西医药杂志,2020,49(19): 2582-2584.
[25] 王燕丽.糖尿病的药物治疗知多少[J].保健文汇,2020(9):2.
[26] SINGH AP, BISWAS A, SHUKLA A, et al. Targeted therapy in chronic diseases using nanomaterial-based drug delivery vehicles. Signal Transduct Target Ther. 2019;4:33.
[27] SOUTO EB, SOUTO SB, CAMPOS JR, et al. Nanoparticle Delivery Systems in the Treatment of Diabetes Complications. Molecules. 2019;24(23):4209.
[28] CADDEO C, PUCCI L, GABRIELE M, et al. Stability, biocompatibility and antioxidant activity of PEG-modified liposomes containing resveratrol. Int J Pharm. 2018;538(1-2):40-47.
[29] 张迪,徐缓,胡美娜,等.脂质体面临的聚乙二醇“窘境”及其解决方法[J].药学学报,2015,50(3):252-260.
[30] DEODHAR S, DASH AK. Long circulating liposomes: challenges and opportunities. Ther Deliv. 2018;9(12):857-872.
[31] NAG OK, AWASTHI V. Surface engineering of liposomes for stealth behavior. Pharmaceutics. 2013;5(4):542-569.
[32] YOSHINO K, TAGUCHI K, MOCHIZUKIET M, et al. Novel analytical method to evaluate the surface condition of polyethylene glycol-modified liposomes. Colloids Surf A Physicochem Eng Asp. 2012;397:73-79.
[33] YAN W, LEUNG SS, TO KK. Updates on the use of liposomes for active tumor targeting in cancer therapy. Nanomedicine (Lond). 2020;15(3):303-318.
[34] XU H, PAXTON JW, WU Z. Enhanced pH-Responsiveness, Cellular Trafficking, Cytotoxicity and Long-circulation of PEGylated Liposomes with Post-insertion Technique Using Gemcitabine as a Model Drug. Pharm Res. 2015; 32(7):2428-2438.
[35] TAKEUCHI I, KANNO Y, UCHIRO H, et al. Polyborane-encapsulated PEGylated Liposomes Prepared Using Post-insertion Technique for Boron Neutron Capture Therapy. J Oleo Sci. 2019;68(12):1261-1270.
[36] HWANG H, JEONG HS, OH PS, et al. PEGylated nanoliposomes encapsulating angiogenic peptides improve perfusion defects: Radionuclide imaging-based study. Nucl Med Biol. 2016;43(9):552-558.
[37] TRUCILLO P, REVERCHON E. Production of PEG-coated liposomes using a continuous supercritical assisted process. J Supercrit Fluids. 2021;167.doi.org/10.1016/j.supflu.2020.105048
[38] 陈刚,王涌臻,唐晓军,等.磁性纳米脂质体在癌症治疗中的应用[J].现代医学,2020,48(9):1234-1238.
[39] 黄海燕.物理化学靶向制剂中磁性靶向研究进展[J].广州化工,2015, 43(1):26-28.
[40] 张辉,胡礼军,喻莉萍,等.芦丁磁性脂质体的制备及质量评价[J].湘南学院学报(医学版),2016,18(1):10-13.
[41] SU W, WANG H, WANG S, et al. PEG/RGD-modified magnetic polymeric liposomes for controlled drug release and tumor cell targeting. Int J Pharm. 2012 5;426(1-2):170-181.
[42] JOSE G, LU YJ, CHEN HA, et al.Hyaluronic acid modified bubble-generating magnetic liposomes for targeted delivery of doxorubicin. J Magn Magn Mater. 2019;474:355-364.
[43] 马秋燕,林华庆,张静,等.磁靶向热敏脂质体在抗肿瘤靶向治疗中的新进展[J].中国医药工业杂志,2019,50(12):1405-1412.
[44] LI Y, XU P, HE D, et al. Long-Circulating Thermosensitive Liposomes for the Targeted Drug Delivery of Oxaliplatin. Int J Nanomedicine. 2020;15:6721-6734
[45] 华雪莲,张树彪,肖义,等.新型阳离子脂质体制备及应用研究[J].化学研究与应用,2019,31(2):177-183.
[46] HE K, LIU J, GAO Y, et al. Preparation and Evaluation of Stearylamine-Bearing Pemetrexed Disodium-Loaded Cationic Liposomes In Vitro and In Vivo. AAPS Pharm Sci Tech. 2020;21(5):193.
[47] HATTORI Y, TAMAKI K, SAKASAI S, et al. Effects of PEG anchors in PEGylated siRNA lipoplexes on in vitro gene‑silencing effects and siRNA biodistribution in mice. Mol Med Rep. 2020;22(5):4183-4196.
[48] 赖可,赵科,刘源,等.空间稳定免疫脂质体的研究近况[J].中国医药工业杂志,2017,48(2):261-266.
[49] 李菲,张娜,郭丰广,等.空间稳定免疫脂质体用于抗肿瘤药物载体的研究进展[J].中国药学杂志,2011,46(24):1865-1868.
[50] 张娟,刘雅利,王旻,等.抗体修饰盐酸抗肿瘤药物脂质体、其制备方法及应用[P]CN201810672398.1,2018-11-06
[51] HAS C, SUNTHAR P. A comprehensive review on recent preparation techniques of liposomes. J Liposome Res. 2020;30(4):336-365.
[52] FILIPCZAK N, PAN J, YALAMARTY SSK, et al. Recent advancements in liposome technology. Adv Drug Deliv Rev. 2020;156:4-22.
[53] CHEUNG CCL, MA G, RUIZ A, et al. Microfluidic Production of Lysolipid-Containing Temperature-Sensitive Liposomes. J Vis Exp. 2020;(157). doi:10.3791/60907.
[54] AHMED KS, HUSSEIN SA, ALI AH, et al. Liposome: composition, characterisation, preparation, and recent innovation in clinical applications. J Drug Target. 2019;27(7):742-761.
[55] LIM CB, ABUZAR SM, KARN PR, et al. Preparation, Characterization, and In Vivo Pharmacokinetic Study of the Supercritical Fluid-Processed Liposomal Amphotericin B. Pharmaceutics. 2019;11(11):589.
[56] MAQBOOL F, MOYLE PM, TAN MSA, et al. Preparation of albendazole-loaded liposomes by supercritical carbon dioxide processing. Artif Cells Nanomed Biotechnol. 2018;46(sup3):S1186-S1192.
[57] VITOR MT, BERGAMI-SANTOS PC, ZÔMPERO RHF, et al. Cationic liposomes produced via ethanol injection method for dendritic cell therapy. J Liposome Res. 2017;27(4):249-263.
|